Bacteria—master compass builders

Compasses have played a pivotal role in the history of navigation. However, for thousands of years before humans invented them, compass needles were manufactured and used for navigation by microscopic bacteria. These bacteria live in almost all aquatic environments around the world.
In his renowned book On the Origin of Species, Darwin stated: “If it could be demonstrated that any complex organ existed which could not possibly have been formed by numerous, successive, slight modifications, my theory would absolutely break down.”1 In line with this, famous evolutionist J.B.S. Haldane claimed that evolution could never produce “various mechanisms, such as the wheel and magnet, which would be useless till fairly perfect.”2 Yet, since Haldane made this claim, both magnets and wheels (circular components of machines that rotate around axles) have been discovered in many species of bacteria, along with many other living things.
How bacteria use them to navigate
It appears that the compass needles passively turn the cells towards North or South, in the same way that man-made compass needles passively turn towards North. However, there is evidence that some species also measure changes in magnetic field strength to optimize magnetic navigation. This makes the system more complex than previously thought.
Why do these bacteria need to know which direction is North? In the water column and sediments there is a layer above which there is oxygen and below which there is almost no oxygen. Magnetotactic bacteria (those that can detect Earth’s magnetic field) like to live in this layer where oxygen concentration is low. If a bacterium swam above this level, the oxygen concentration would be too high and it would need to swim back down. This is where the compass needles come in.
Magnetic field lines are best known to travel from the earth’s magnetic South Pole to the North Pole. But they also come up out of the earth in the Southern Hemisphere and bend back down into the earth in the Northern Hemisphere (fig. 1). Thus, bacteria wanting to travel downwards travel towards magnetic North in the Northern Hemisphere, and towards magnetic South in the Southern Hemisphere. If these bacteria are moved between hemispheres, they soon learn that the downwards direction is now in the opposite direction relative to the magnetic field. The bacteria can also find optimal growth conditions more quickly because they only travel forwards and backwards along the magnetic field lines, rather than in a random three-dimensional swim.
Such compasses are just one component of these microbes’ complex navigation system. A cell’s navigation system (which includes short-term memory) processes thousands of signals from sensor arrays that detect different aspects of its surroundings. The output signals are then relayed to the cell’s motors (see ‘Amazingly designed motors’, p. 52) to steer the cell towards optimum growth conditions.
Compass construction
Building the compass needles within the bacteria is no small engineering feat. It requires an exceptionally high level of control and coordination by a number of molecular machines. The needles are made of magnetic crystals of the mineral magnetite (iron oxide Fe3O4), or sometimes greigite (iron sulfide Fe3S4).

The process begins with a bacterial cell constructing numerous ‘reaction chambers’ called magnetosomes. Each one is made by folding the inner membrane of the cell inwards to form a spherical compartment (like a bubble). Through a complex production line, machines in the cell manufacture specialized proteins according to instructions on DNA. These proteins are then sorted and inserted into the magnetosome membranes. They are then modified by more machines in a coordinated stepwise manner. The proteins connect to form larger structures which function as molecular devices that keep conditions inside the magnetosomes within finely-tuned parameters. This is necessary to support crystal growth. In particular, they regulate the iron level, the redox (reducing/oxidizing) potential, and the pH (acid-base balance).
The cell harvests iron from the surrounding environment, importing it through a complex system of sensors and molecular machines. This includes a tiny electric motor which powers the iron-transport machinery. Once inside, iron must be handled carefully as it is toxic. Bacteria have complex, highly regulated systems to manage this risk. They store iron in special protein containers until it is needed (fig. 2). Machines chemically modify the iron for storage before packing it inside the containers.
Iron is then transported inside the magnetosomes to begin crystal formation. Crystal growth appears highly regulated so that only one crystal grows inside each magnetosome. As the crystals grow, over ten different types of proteins fine-tune their size and shape, making them highly uniform.
Different species make crystals of different sizes and shapes. The number of crystals per cell also appears to be tightly regulated. Enough crystals need to be made to form sufficiently strong compass needles, but creating extra would be a waste of energy. This intricate regulation results in crystals of exceptionally high quality, with strong magnetic fields and a uniform size distribution.3 This makes them “increasingly sought after … for industrial and medical applications.”4 Humans have struggled to produce magnetic nanocrystals of such high quality.

Finally, the crystals are assembled into one or multiple compass needles (fig. 3). Long cable-like fibres are constructed from one end of the cell to the other to form a “sophisticated mechanical scaffold”5 called the magnetoskeleton. In a coordinated process involving yet more machinery, the magnetosomes are attached to the magnetoskeleton and moved along it to form a long chain of crystals. The magnetoskeleton must have a strong design to stop the needle from bending and collapsing in on itself due to the magnetic attraction between the crystals. The completed compass needle can now function as “an extremely efficient magnetic sensor”.4
The cells of some bacteria are helical in shape, like a corkscrew (fig. 3). Constructing a straight compass needle inside these cells, tethering it in position, and perfectly aligning the needle with the cell’s swimming axis is quite an engineering challenge. Yet, these bacteria can do it. Just how they do it is the subject of more research.3 If the needle were not aligned correctly, the cell would tumble as the swimming and magnetic forces would pull it in slightly different directions.
Separation of magnetosomechains during cell division
When bacteria reproduce by dividing into two new cells, their compass needles must be cut and equally divided between the two cells. The needles are positioned perfectly at the division site, cut with the “highest possible precision at midchain” and separated into the two cells with “unexpectedly high accuracy”.6 The two half-needles are magnetically attracted to each other, which makes separating them another engineering challenge these bacteria must overcome. They appear to do this using a leverage mechanism which requires the coordination and use of yet more machines and structural scaffolds. The two half needles are grown to full needles and transported to the middle of each of the two new cells. How the needles are transported and stop once perfectly in position is still unknown, but appears highly regulated.
Magnetic symbiosis
Protists are a large group of single-celled organisms which, unlike bacteria, have a nucleus—for example, amebae. One species of protist that uses flagella (with a radically different and even more complex design than bacterial flagella) to swim attaches a species of non-swimming magnetotactic bacteria to itself. The surface of this protist is covered with many of these bacteria, which all align themselves with the earth’s magnetic field. This in turn aligns the protist with the field, the two species becoming a magnetic superorganism, in effect. The protist benefits by being able to navigate along Earth’s field. The bacteria also benefit, since they can’t swim by themselves. The two species also trade some metabolic products.
Monteil, C. and 15 others, Ectosymbiotic bacteria at the origin of magnetoreception in a marine protist, Nature Microbiology 4:1088–1095, 2019.
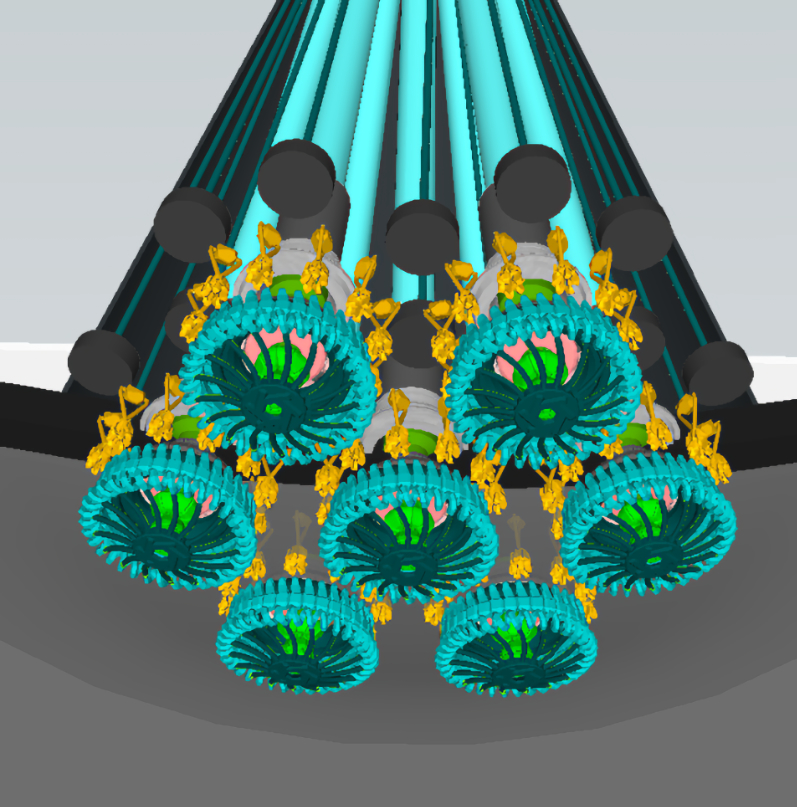
Amazingly designed motors
Magnetotactic bacteria swim using sophisticated electric motors which rotate long helical propellers called flagella. These motors have many parts which perform similar functions to parts in human-designed motors. These parts include gears, rotors, axles, driveshafts, bushings, ball bearing-like bearings, oil-like (lipid) lubricant, structural scaffolds, hinges, universal joints, other joint types, adaptor rings, switches, stators, capacitors, and sensors.1 One magnetotactic species has one of the most complex and powerful engines in living things, illustrated here. This engine consists of multiple coordinated motors,2 and enables the bacterium to swim at over 200 body lengths per second.3 Like the compass needles, these electric motors and their factory-like programmed construction present a profound obstacle to the idea that they just arose from unintelligent matter.
- Thomas, D., E. coli’s electric motor: a marvel of design, Creation 44(1):42–45, 2022; creation.com/e-coli-motor.
- Ruan, J., and 8 others, Architecture of a flagellar apparatus in the fast-swimming magnetotactic bacterium MO-1, pnas.org, 11 Dec 2012. See also creation.com/7motors1.
- Bente, K. and 7 others, High-speed motility originates from cooperatively pushing and pulling flagella bundles in bilophotrichous bacteria, elifesciences.org, 18 Jan 2020.
Conclusion
Bacteria are far from ‘primitive’. They can manufacture compass needles of exceptionally high quality and use them to navigate along magnetic field lines with complex electric motors. There is much still to be learnt about these incredible navigational devices. As one researcher said, “[bacterial magnetic navigation] still bears unexpected and sophisticated features waiting for discovery.”7 Such discoveries will no doubt continue to reveal God’s creative intelligence and be even more difficult to explain via evolution.
References and notes
- Darwin, C., Origin of Species, New York University Press, 1872. Return to text.
- Is evolution a myth? A debate between D. Dewar and L.M. Davies vs J.B.S. Haldane, Watts & Co. Ltd / Paternoster Press, 1949. See also Thomas, B., Virus motors impossible for evolution, icr.org, 9 Jan 2009. Return to text.
- Müller, F. and 2 others, A compass to boost navigation: Cell biology of bacterial magnetotaxis, J. Bacteriol., journals.asm.org, 8 Oct 2020. Return to text.
- Moisescu, C. and 2 others, The effect and role of environmental conditions on magnetosome synthesis, Front. Microbiol., frontiersin.org, 11 February 2014. Return to text.
- Toro-Nahuelpan M. and 7 others, MamY is a membrane-bound protein that aligns magnetosomes and the motility axis of helical magnetotactic bacteria, Nat. Microbiol., nature.com, 29 Jul 2019. Return to text.
- Toro-Nahuelpan M. and 5 others, Segregation of prokaryotic magnetosomes organelles is driven by treadmilling of a dynamic actin-like MamK filament, BMC Biol., bmcbiol.biomedcentral.com, 12 Oct 2016. Return to text.
- Müller F., Perfect navigation: How to fit a compass needle into a helical bacterium?, naturemicrobiologycommunity.nature.com, 2 Aug 2019. Return to text.








Readers’ comments
Comments are automatically closed 14 days after publication.