Feedback archive → Feedback 2009
Dino proteins and blood vessels: are they a big deal?
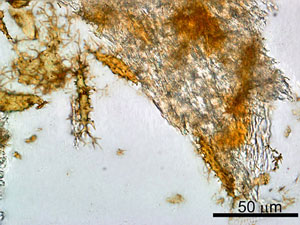
Scott W from the US writes in connection with our recent article Dinosaur soft tissue and protein—even more confirmation!
I wonder if you could comment on the following blog perspective on this recent find:
I know lots of creationists that get charged up by this sort of thing, since this is supposed to be evidence that the fossils must be younger than conventional claims by millions of years. I’ve never been very excited about this, mainly because the Flood throws a real monkey wrench into these arguments. A major source of degradation of biomolecules is water. So anything that died in the Flood and floated in water for a year shouldn’t really have many biomolecules left, right? Seems like if you believe fossils are millions of years old or are the remains of carcasses from the Flood, the outcome is the same: very little preservation of biomolecules. In this case, Schweitzer is finding collagen, which is pretty tough, so I guess it makes sense that it might be preserved in some extraordinary cases. In any case, finding the rare, hardy biomolecule from dinosaurs is nothing to get excited about, since the conventional model and the Flood model both predict the same thing: this should be very, very unusual.
I think it’s still FAR more plausible for this organic material to survive water than billions of years, but would love to hear your comments. Perhaps you could add an ‘update’ to the article.
Drs Carl Wieland and Jonathan Sarfati reply:
Sure thing.
This blogger must be about the only one, creationist or evolutionist, who does NOT see this as a problem for the millions of years. Why else would Dr Schweitzer say she redid one experiment 17 times before she would believe the evidence of her eyes?1 Why would so many others express disbelief that these structures could have survived so long, and why would theories of desperation like biofilms be invented?
We have never heard any creationist geologist suggesting that all fossils floated for a year before being buried. Far from it. Many of the fossils, such as fossil fish, show remarkable preservation consistent with rapid burial.2 In a complex set of interconnected catastrophes such as the Flood, one would expect rapid burial by all sorts of sedimentary activity which may be at any stage of the flood—turbidity currents (submarine landslides), for example, would have been capable of burying creatures, including even in the very early stages of the Flood.
Even if the dinosaur carcass were exposed for some weeks or months before being buried, a creature of such significant size would by no means be exposing all its organic constituents to degradation, whether by oxygen, bacterial action, or water. Collagen fibres are often notable on marine carcasses of large creatures, where the creature as a whole has obviously been decomposing on a beach, exposed to water at every high tide, for months. This is not a surprising, or even controversial, observation.
The most likely situation in this sedimentary sandstone which the reports state encased this particular dinosaur carcass is that the sandstone was deposited over a relatively brief period of time and that the creature died not long before, or even as, it was encased in sediment, which subsequently hardened.
To say that both models predict the same thing really surprises us. The author does not seem to engage with (or is unaware of) the statements in the articles on our site (and those we cite from secular journals) about such fragile molecules falling apart, even if hermetically sealed, from thermodynamic considerations alone. The difference in the predictions is stark—even the most optimistic predictions had collagen not surviving even 3 million years at freezing point, and less than 200,000 years at only 10ºC and less than 15,000 years at 20ºC (in general, reaction rate increases exponentially with temperature3, and dinosaurs are supposed to have lived in a warm climate).
But to explain further why this blogger is in error requires some fairly basic physical chemistry (the speciality of the second author JS) and organic chemistry (which he also studied extensively).
In aqueous solutions, water is so much more concentrated than anything else, and doesn’t change very much (~55.5 mol/kg) so that its activity is treated as constant when it comes to equilibria. So the extra water at the time of the flood would barely affect equilibria; ANY aqueous solution would have so much excess water that the reaction would be driven to completion. The only time water activity is counted is when the mixture is almost dry so that water concentration differences then matter.
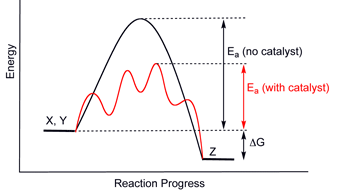
But most important is the reaction kinetics, i.e. the rate that the system heads towards equilibrium. The thermodynamic equilibrium depends only on the free energy differences between starting and end materials (reactants and products), while kinetics (reaction speed) depends on the free energy differences between starting material and the transitional state or reaction intermediate (i.e. activation energy). In simple terms, for the reaction to occur, some bonds of the reactants must be broken, and this takes energy. To apply this: a diamond exposed to the air at room temperature is very far from equilibrium (which would be to form CO2), but the reaction is too slow to measure because it requires a very high activation energy to break the very strong bonds [diamond in isolation is not at equilibrium either, but again conversion to graphite is too slow to notice].
Protein hydrolysis occurs very slowly, even though the equilibrium is far towards the monomers, because the activation energy is too high. But as research showed, the reaction would be completed in well under 70 million years. And again, in an already aqueous solution, extra flood water would make no difference.
What does make a difference is pH: hydrolysis occurs much faster if catalyzed by acid or alkali (that’s why these are so corrosive on the skin, which is made of the protein keratin). Note that a catalyst doesn’t change the equilibrium but provides a different intermediate pathway with a lower activation energy, speeding up the reaction exponentially (see diagram). For example, an experiment showed “reaction rate increased proportionally with hydroxide ion concentration”,4 that is, with increasing pH or alkalinity.
So the worst thing for hydrolysis is a neutral pH (~7), neither acid (pH < 7) nor alkaline (pH > 7).5 And ground water and geothermal waters are usually neutral. Not only that, they are often neutral buffer solutions. A buffer contains weak acidic and alkaline components that react with added strong acid or alkali, so tends to resist pH changes. Groundwaters tend to dissolve both carbonates (weak alkalis) and CO2 (weak acid), which is why they are buffers6 —blood likewise has this buffer solution to keep it within a narrow pH range, in this case very weakly alkaline.7 So these neutral-buffered waters would serve to retard acid or alkaline hydrolysis, contrary to this blogger’s claims.
So the proven rules of chemistry show that both mainstream creationists and mainstream evolutionists are right to see these dino remnants as a big deal. But for evolutionists, it’s a problem to be explained away, while it’s a welcome development under a biblical framework.
References
- Mary Schweitzer, Science 307:1852, 25 March 2005. Return to text.
- Daniel Woolley, Fish preservation, fish coprolites and the Green River Formation, J. Creation 15(1): 105–111, 2001. Return to text.
- This is the simple Arrhenius rate equation k=Aexp(−Ea ∕RT), where k is the rate constant, A is a temperature-independent constant (often called the frequency factor), exp is the function ex, Ea is the activation energy, R is the universal gas constant, and T is the absolute temperature. Return to text.
- Bruce Robinson and Jefferson Tester, Kinetics of alkaline hydrolysis of organic esters and amides in neutrally-buffered solution, International Journal of Chemical Kinetics 22(5):431–448, 30 Sep 2004. Return to text.
- For the chemically minded, pH ≈−log10[H+] (square brackets mean concentration), and in water [H+][OH–] = 10−14. So neutral water has a minute concentration of both H+ and OH–, both 10−7 M (at 25°C). Return to text.
- Kirk Nordstrom, Pre-mining ground-water quality in debris fans developed from pyrite-mineralized erosional scars in the Red River Valley, New Mexico: comparison between mined and unmined areas, Geological Society of America Abstracts with Programs 37(7):395, 18 October 2005: “Ground waters in bedrock are of neutral pH, buffered by dissolution of carbonate minerals … .” And Ref. 4 refers to “… neutrally-buffered solutions such as many groundwater and geothermal fluids.” Return to text.
- The pH of a buffer depends on the acidity components and proportions. For the chemically minded, use the Henderson–Hasselbalch equation pH = pKa + log10([conjugate base]∕[acid]), where pKa is −log(Ka) where Ka is the acid dissociation constant. Return to text.



Readers’ comments
Comments are automatically closed 14 days after publication.