Journal of Creation 28(3):73–79, December 2014
Browse our latest digital issue Subscribe
Heredity is foundationally cellular, not genetic, and life’s history is discrete, not continuous
Genes carry some characteristics of life, but life itself is inherited only via whole functional cells—essential elements of the mother’s functional body plan (a design that works). If this maternal foundation is disrupted by mutation, the embryo dies and it is not passed on. The history of life is therefore not an evolutionary continuum, like a single tree as Darwin imagined, but a forest of created kinds, each with its own unchanging functional body plan. The created kinds diversified into today’s species via conserved core processes that are built in to the functional body plan. They only come into play late in embryogenesis, after the functional body plan has been established by the mother’s egg cell. Evolutionists have assumed (without explanation) the existence of life and its variable reproduction, and this oversight has misled them into a grand delusion about the history of life on Earth. To understand life’s true history we need to locate the boundaries between unchanging body plans and diversifying mechanisms during embryogenesis.

After Kirschner and Gerhart,5 p. 268.
Click for larger view.
For over a century now the subject of heredity has been dominated by Mendel’s discovery that some characteristics of life are inherited in particulate form via what we now call ‘genes’. Geneticists have ever since been totally focused upon the inheritance of such characteristics and they have entirely overlooked the main issue—the inheritance of life itself. This obfuscation of heredity led to the debacle of neo-Darwinian theory, now exposed as having been vacuous from its beginning.1 In this article, I show from the mechanical logic and molecular detail of reproduction that the inheritance of life is carried via cells, not genes, and that the history of life has been discrete, not continuous (as Darwin and his supporters have erroneously assumed.)
The nature of inheritance
In R.A. Fisher’s 1930 book that launched the Modern Synthesis (neo-Darwinian theory) The Genetical Theory of Natural Selection, the first chapter dealt with the nature of inheritance. Fisher compared Mendel’s particulate theory of inheritance with Darwin’s blending theory. He considered a wide range of evidence supporting particulate inheritance and confidently concluded that it “seem[ed] capable of excluding the possibility that blending inheritance can account for any appreciable fraction of the variance observed”.2 Fisher’s focus upon “variance” ignored its complement—invariance. He was not alone. It seems that everyone who has ever considered heredity has always looked at variation rather than ‘sameness’. Yet sameness is overwhelmingly the most prominent, obvious, and inescapable conclusion that every scientist is confronted with when studying heredity. If we were to add up all the breeding experience that we have accumulated over thousands of years with domesticated species, plus all the experimental data documented over the last century and a half on crop plants, fruit flies, bacteria, yeast, worms, fish, frogs, mice, etc. then the clearest possible conclusion would be sameness. Wheat produces wheat, worms produce worms, and mice produce mice. Sameness is the ‘elephant in the room’ on the subject of heredity.
How does life manage to stay the same when all around (and within) is changing? The closest I have come to finding this matter dealt with in the scientific literature is the 1997 book by Berkeley Professor John Gerhart and Harvard Professor Marc Kirschner entitled Cells, Embryos, and Evolution: Toward a cellular and developmental understanding of phenotypic variation and evolutionary adaptability. They outlined an evolutionary paradox that molecular biology had uncovered. On one side there is extraordinary conservation of molecular structure and function in the core processes of life. All prokaryote cells have similar structure and core metabolic machinery. All eukaryote cells have similar structure and core metabolic machinery (prior to their developmental specialization). All multicellular animals (they only considered animals) use the same intercellular junctions and extracellular matrix. Body plans remain exactly the same across entire phyla. Information processing is so highly conserved that a human insulin gene can be inserted into a bacterium or yeast genome and exactly human insulin is produced.
In contrast, there is extraordinary diversity in the anatomy, physiology, and behavioural strategies among individual species. How can so many things that remain the same produce so many ways of being different? Neo-Darwinian theory has no answer to this paradox because it depends entirely upon random DNA copying errors for producing novelty, and as a result evolutionary biologists had expected to find random changes in everything! They were clearly wrong.
In their next book, The Plausibility of Life: Resolving Darwin’s Dilemma, Kirschner and Gerhart did resolve the paradox in what they claimed to be the first comprehensive theory of how life works at the molecular level: facilitated variation theory.3,4 They identified “weak regulatory linkage” between “modular conserved core processes” as the solution. They compared the modular components to Lego blocks—individually hard to break (and thus conserved), but easy to pull apart and rearrange (weakly linked) to make different numbers, kinds, sizes, and shapes of organs and organisms. Furthermore, they claimed that adaptability, and thus ‘evolvability’ is built in. Genetic variation and mutations do not have to be creative, they only need to trigger the creativity built in to every organism.5
Biological functionality
Just as biologists have overlooked ‘sameness’ in searching for a theory of inheritance, they have also overlooked life’s functionality. This is an absurd situation because functionality is fundamental to life. Evolutionists just assumed without explanation that organisms can survive and reproduce themselves in variable forms. This is such a stupendous accomplishment that it needs to be explained for its own sake before any of its consequences—such as ‘evolution’—can be discussed with either clarity or credibility.
Biophysicist Howard Pattee identified this problem over 40 years ago:“… if you ask what is the secret of life, you will not impress most physicists by telling them what they already believe [—] that all the molecules in a cell obey all the laws of physics and chemistry. The real mystery, as in any machine, is in the origin of the highly unlikely and somewhat arbitrary constraints which harness these laws to perform specific and reliable functions. … Function is a process in time, and for living systems the appearance of time dependent function is the essential characteristic of [its] hierarchical organization. … [I]t is easy to understand how a simple change in a single variable can result in very complicated changes in a large system of particles. This is the normal physical situation. It is not easy to explain how complicated changes in a large system of particles can repeatedly result in a simple change in a single variable. It is this latter result which we interpret as the ‘integrated behavior’ or the ‘function’ of a hierarchical organization. Thus, we find structural hierarchies in all nature, both living and lifeless, but functional hierarchies we see as the essential characteristic of life, from the enzyme molecule to the brain and its creations [emphasis in original].”6
Kirschner and Gerhart’s ‘conserved core processes’ identify exactly those components of life that make it functional and keep it functional over thousands of years against the natural tendency of all things to decay.7 Human design engineers know from experience that only some combinations of machine components and only some methods of operation will produce a desired outcome. An airplane, for example, consists of numerous parts, none of which can fly. Only when all the right parts are appropriately put together and only when the machine is operated in the correct manner will the airplane fly. In similar manner, biologists have proved beyond any doubt, time after time, that life only ‘works’ in cellular form.
Consider this recent description of the LUCA (the supposed last universal common ancestor):
“… the modern view shows that LUCA is not [Darwin’s] ‘primordial form’, but rather a sophisticated cellular organism that, if alive today, would probably be difficult to distinguish from other extant bacteria or archaea.”8
If the ancestor of cells was a cell, then cells clearly are the carriers of life. World-leading researcher on the origin of life, Harvard’s Jack Szostak, has narrowed his search down to the origin of the first cell:
“We assume we have the chemical building blocks of life: the question we are looking at is what do we need to do to make these chemicals get together and work like a cell?”9
Evolutionists are here providing ‘hostile witness’ to the fact that life works only in cells, and life can only be passed on in cells. Every biologist has known this ever since Louis Pasteur established the principle that ‘life comes from life’ in 1864!10
The regulatory genome
Kirschner and Gerhart recognized cells (and their persisting functionality) as conserved core processes, while diversification is achieved via intergenerational changes in regulatory circuits. The nature of these regulatory changes has been beautifully illuminated by the work of developmental biologist Eric Davidson and physicist Roy Britten.11 They have now identified the gene regulatory networks (GRNs) that control early embryo development in the purple sea urchin,12 for which Davidson won the 2011 International Prize for Biology.13 “GRNs are composed of multiple sub-circuits, that is, the individual regulatory tasks into which a process can be parsed are each accomplished by a given GRN sub-circuit.”14 Four different kinds of modules appear in their GRNs: ‘kernels’, which lie at the core of any control system; ‘plug-ins’, which are modules that accomplish particular functions and can be found at many different intermediate levels of operation; ‘I/O units’, which allow communication and transport (inputs and outputs) between modules; and ‘differentiation gene batteries’, which turn different cell types into their final form in the late stages of embryo development.
Davidson’s GRNs operate under three levels of hierarchical control. At the top level are the body plan kernels, which tolerate no change (mutants die) so they are the same across any Phylum (figure 1). Second-tier controls develop the organ and appendage systems within the body plan. They consist of plug-ins and I/O units and are highly conserved (some change may rarely be tolerated) and they correspond approximately to the Class, Order, and Family levels in taxonomy. But third-level controls—the differentiation gene batteries—tolerate possibly endless change, and this is where Davidson believes that most species- and genus-level variation takes place.15, 16 This hierarchical structure dramatically constrains the impact of mutations because their effects will depend upon the level of the hierarchy in which they occur.
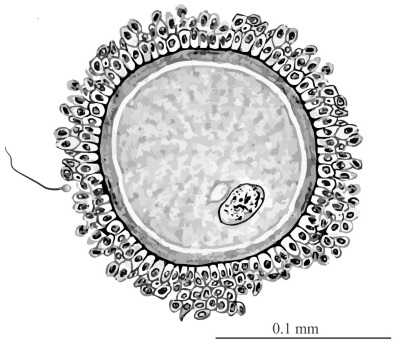
Figure 2. Human egg (ovum) with sperm (on left side). The ovum is surrounded by a protective coating of ordinary-sized cells. Click for larger view.
The invariance of body plans across phyla poses a severe challenge to evolution:
“A strong conclusion is that the evolutionary process generating the … basic eumetazoan body plans was in many ways very different from the evolutionary changes that can be observed at the species level in modern animals [emphasis added].”17
Creationists may recognize this statement as a biological description of Genesis 1, where God created the various kinds of life to reproduce “according to their kinds”. Evolutionists have no credible explanation for what Davidson is describing here.
Molecular mechanisms of stasis
According to Davidson, stasis is achieved at the molecular level through “recursive wiring” of kernels and regulatory “lockdowns” during embryo development. ‘Recursive wiring’ means that kernels contain information that is required at multiple stages during embryo development. The development process must refer to the kernel over and over again and as a result any change in the kernel is “disastrous”.15
‘Lockdown’ during embryo development is a switching operation. For example, once the head–tail, left–right, and front–back axes of an embryo are established, new embryonic cells move into these regions and begin multiplying to build the relevant organs and appendages within the body plan. Every cell in each region (territory) of the body plan will have these axis-oriented circuits ‘locked down’ so they cannot change at any later stage. Consequently, every new cell that goes towards building a leg, for example, will build a ‘right front leg’ in the right front position, a ‘left front leg’ in the left front position, and so on. Only if these foundational body plan circuits are locked down and unable to be changed at any later time will the embryonic process lead to a functional outcome.
The mechanical logic of reproduction
The mechanical logic of reproduction establishes beyond any doubt that heredity is carried by cells. Single-celled organisms are given a whole share of their parent’s life by the parent cell simply dividing in two. If one (or both) of these daughter cells fails to function there is no inheritance. A similar principle applies to asexual reproduction in multicellular organisms. In sexually reproducing organisms the mother’s egg cell provides the necessary foundation for life in the offspring. Fathers only contribute half of the necessary DNA. We are discovering an increasing number and range of multicellular species (including vertebrates) where the female deletes the father’s contribution and reproduces via parthenogenesis, either sometimes, or always.18 All of these mechanisms are equivalent in that the inheritance of life in all species requires a functional whole cell, not just a packet of genes.
This mechanical logic is on display in a comparison of the human egg (ovum) and sperm in figure 2. The egg is about a thousand times larger than the sperm, and is surrounded by protective layers of ordinary-sized cells. The tiny sperm head is of comparable size to the nuclei of the cells surrounding the ovum because it only contains chromosomes (a half set) plus some mitochondria to provide energy in the race to succeed during fertilization. In contrast, the ovum (including its nucleus) is hugely swollen over normal-sized cells because it is packed with all the normal cell requirements (molecular machinery for survival and variable reproduction) plus the necessary extra RNA, ribosomes, and proteins required to establish the body plan of the offspring and to launch the offspring’s unique new genome into successful operation. No researcher would try to produce an embryo from sperm alone because we all know it wouldn’t work. In contrast, laboratories around the world are doing reproductive experiments with egg cells of all kinds, because they know that the cell—and nothing less than the cell—is the fundamental requirement for successful reproduction. In plants, just one cell from a meristem region of the largest tree can be grown in laboratory culture and then induced (with just two hormones) to turn into another great tree of exactly the same kind. In zebrafish, and possibly all other animals, just two opposing ‘morphogens’ are all that is required to launch a fertilized egg onto its complex developmental journey towards adulthood.19 Not a single species on Earth can produce offspring from a sperm (even though it contains the necessary DNA), yet a single ovum in a parthenogenetic species can do so. QED. Heredity is carried via cells, and nothing less!
The molecular logic of reproduction
There is a compelling logic in the molecular details of cellular inheritance, just as there is in its mechanical details. Fruit flies were early subjects of reproductive experiments and German embryologist Christiane Nusslein-Volhard won a Nobel Prize for her work on the genetic control of fruit fly development. Her 2006 book Coming to Life: How genes drive development tells the story in easy-to-understand words and drawings.20 Her experimental method involved exposing fruit flies to mutagenic chemicals so that they produced deformed embryos. She then tracked down which genes had been damaged and how this damage affected development. After a great deal of trial and error she produced a list of genes essential to early embryo formation, together with descriptions of the consequences of damaging them. She also exposed fertilized eggs to mutagens so that mutations in the zygotic genome could be distinguished from mutations in the maternal genes. Her story only covers the very early stages of embryonic development because the mutant embryos died before they could reveal what effects their damaged genes might have had further downstream. This discovery is fully consistent with Eric Davidson’s finding that regulatory kernels are recursively wired and any change to them is disastrous.
Nusslein-Volhard says, “If all cells [of the embryo] have all genes [of the organism], the origin of differences arising during development of an organism must reside in the cytoplasm.” This principle of the egg having an ‘intrinsic polarity’ was established by Theodor Boveri in the 19th century.20 In the fruit fly, before fertilization occurs, and even before the mother’s egg cell is released from its ovary, the head–tail axis, the left–right axis, and the front–back axis of the potential offspring are already established and they are associated with a set of four maternal gene groups. She says:
“From there, the cues provided by these [four] maternal gene groups [originating in the egg cell] spread out and organize the pattern of the entire embryo. … Mutations of maternal genes lead to particularly dramatic changes in the shape of the larva. This makes perfect sense because maternal genes affect the earliest functions of embryo pattern formation. For example, embryos from mutant female flies lack large body regions. … Bicoid embryos lack all structures that would normally result from the front half of the egg, oskar embryos lack the abdomen, and in the case of torso both the very front and the very back are missing. There are several genes with phenotypes like oskar or torso. … A fourth maternal gene group determines the dorsal-ventral [front-back] axis … and most mutants lack all of the ventral [front] side such that the dorsal [back] structures of the embryo continue all the way around its circumference [emphasis added].”20
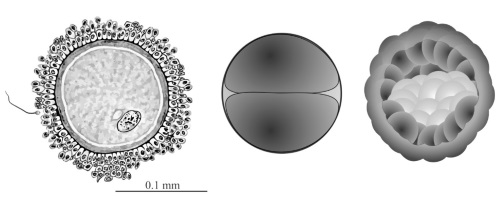
Figure 3. Early human reproductive stages. Human egg and sperm (left) for size comparison; the first zygotic cell division (centre); blastocyst stage interior view (right) at day 5 after fertilization showing the inner cell mass (ICM, light-coloured cells) that will become the embryo. Click for larger view.
No wonder the poor mutants died!
Eric Davidson generalized this polarization (prepatterning) of maternal egg cells to all animal clades except mammals.16. However, Gardner had previously challenged this widely cited exception on the grounds that it had been poorly researched, and he demonstrated conclusively that “the mammalian egg is unquestionably a polarized cell”.21 Immediately upon fertilization in the mouse we see the egg nucleus firmly in control. Its machinery unpacks the paternal genome and repackages it to form the ‘paternal pronucleus’.22 The maternal genome is also reawakened and repackaged into a ‘maternal pronucleus’. Both genomes then get ready for the all-important step of joining up prior to the first cell division. The first division of the human zygote, illustrated in figure 3, generates one cell that will later produce the embryo, while the other cell produces the embryo-support structures including the placenta. The blastocyst stage, 5 days post-fertilization, illustrates this (figure 3, right). The outer layer of cells goes on to produce the placenta, but the inner cell mass (ICM, light-coloured cells) remains in an entirely quiescent and pluripotent state until the whole is implanted in the uterus wall. Only after implantation will the ICM begin to differentiate into the new offspring. However, even after activation of the embryonic genome, successful development of the embryo relies on stored maternal components, without which it fails to progress beyond the initial cell divisions.23
The maternal-to-zygote transition (MZT)
The molecular logic of cellular heredity is best illustrated by the hand-over of control in animal reproduction from the mother to the offspring—technically called the maternal-tozygote transition (MZT). Similar principles apply in plants24 but I have not reviewed the situation in fungi.
The mother’s egg and the father’s sperm are very specialized gametes. When they join up during fertilization they need to quickly lose all of their specializations as gametes so that the fertilized egg can take on a totipotent state and be ready to become the first cell of the new offspring. To achieve this, the entire zygotic genome is shut down. This is achieved by a number of epigenetic changes, including the removal of DNA-methylation, histone modifications, and chromatin remodelling. Some markers may evade shutdown, thus producing a means of ‘epigenetic inheritance’. These include ‘imprinted’ or silenced genes that might otherwise interfere with development, X-chromosome inactivation in order to achieve correct female gender identity, and marks that allow parent-specific inheritance of some features, including disease.25,26 During this period of zygotic genome shutdown the mother’s RNAs in the cytoplasm and nucleus direct the operations of the cell, and her ribosomes make the proteins that are required during the transition period. Then when the time is right to make the transition, the maternal RNAs begin to break down and transcription starts up from the zygote’s genome.
A review of the MZT in animals described it in this way:
“The maternal genome controls virtually all aspects of early animal development. Maternal mRNAs and proteins, which are loaded into the egg during oogenesis, implement basic biosynthetic processes in the early embryo, direct the first mitotic divisions, and specify initial cell fate and patterning. As development proceeds, two processes are triggered that together form the maternal-to-zygotic transition (MZT): first, a subset of the maternal mRNAs is eliminated; second, the transcription of the zygotic genome begins. Initially, the destruction of maternal mRNAs is accomplished by maternally encoded products. However, zygotic transcription leads to the production of proteins and microRNAs (miRNAs) that provide feedback to enhance the efficiency of maternal mRNA degradation [emphasis added].”27
Changeover must be highly coordinated to ensure the functionality of the embryo at all times. And, as noted earlier, the zygotic genome still needs a cache of stored maternal components for a successful outcome.5 These concurrent requirements place very tight constraints on development and demonstrate pervasive maternal control.
Nusslein-Volhard described the impact of mutations in the zygote’s genes (zygotes that were exposed to mutagens):
“There is more variety among the phenotypes of [mutated] zygotic genes. The missing regions are usually smaller, and the defect is visible only after gastrulation” (p. 53).
Gastrulation is the stage following the blastocyst (figure 3, right) when embryo structure begins to form. Prior to gastrulation the mother establishes what Kirschner and Gerhart call the ‘invisible anatomy’ of the body plan (figure 1) in each of the blastocyst cells. At gastrulation, these cells begin to differentiate into the various regions and organs of the new body according to the mother’s plan. Nusslein-Volhard is telling us that, unlike mutations in maternal genes—which cause deletion of large body regions—mutations in the zygote genome only produce smaller ‘missing regions’ and they appear only during the structure-forming stage of embryo development, and not before. The zygote genome has no control over the laying down of its own body plan!
The formation of primordial germ cells (PGCs) in Drosophila provides us with a magnificent illustration of both cellular inheritance and MZT in the one phenomenon. PGCs are formed very early in the Drosophila embryo and are then held separate from the somatic cells that produce the body. The PGCs remain quiescent until it is time for reproductive development in the adult fly, when they ‘wake up’ and go on to produce the gametes for the next generation. The mother thus places the germ cells for her grandchildren in a safe place within her child until it is time for them to develop! This means that her child has no control over the cellular heredity of the grandchildren. By reaching across a generation in this manner the mother prevents any possibility of ‘evolution on the grand scale’. This is an extra fail-safe method of guaranteeing functional stasis, generation after generation.

Figure 4. Summary of the maternal to-zygote transition (MZT) in Drosophila. In somatic cells (left) degradation of maternal RNA is initiated by maternal factors (a), then zygote-mediated decay begins (b), and finally takes over (c). Zygotic transcription (dotted line) maximizes at 3.5 hours, while the MZT (heavy dash/dot line) occurs at around 2.5 hours. In PGCs (right) the transition is sharper and the MZT occurs later at about 4 hours, well after the PGCs have formed at 1.5 hours. As a result, no zygotic decay factors are present in the PGCs and they remain intact and dormant until sexual maturity. (After Simonelig.28) Click for larger view.
Figure 4 shows the decay of maternal mRNA and increase in zygotic mRNA, for both somatic cells and PGCs, over the first 6 hours after egg deposition in Drosophila. The MZT in somatic cells (left) occurs during the third hour, and maternally initiated decay of maternal RNAs is almost complete by the end of the third hour. In contrast, the PGCs (right) are produced at around 1.5 hours and the MZT does not occur until 4 hours.28,29 In this way the mother retains full control of her child’s reproductive cells (which will produce her grandchildren) until the point where the remainder of her child’s body is fully under its own control. It is therefore impossible for the child to change the functional body plan handed down through it to her offspring.
In mammals the separation between embryo and support cells starts with the first cell division. In humans, zygotic gene expression at this stage only concerns the cell-division cycle and maintaining pluripotency of the daughter cells.30 Differentiation does not appear until the blastocyst stage (figure 3) when the inner cell mass (ICM) is distinguished from its outer covering (which forms the placenta after implantation in the uterus). The ICM is maintained in “an entirely pluripotent and unspecified state”16 for over a week before the MZT allows the zygotic genome to begin building the new offspring’s body from the ICM.31 The mother thus ensures that her functional body plan has been established before she allows her offspring to begin its own individual development.
Conclusions
Life is passed on from one generation to the next in whole cells, not just in genes or genomes. It is passed on in toto in the egg cell, in an all-or-nothing, fail-safe transaction between mother and offspring. The mother’s functional body plan (a design that survives and reproduces itself with variation) is built into the offspring’s body during the early stages of embryogenesis when mutations are lethal. The zygote’s genome takes over control only after the mother’s body plan has been put into place. Variations within the body plan only become possible during the later stages of embryogenesis. The boundaries between the unchanging and the variable stages will require much more research at the molecular level. The unalterable nature of the functional body plan is a discovery that destroys the foundations of ‘evolution on the grand scale’. Life on Earth does not constitute a single evolutionary tree, as Darwin imagined, but a forest of created kinds and their descendants.
Acknowledgments
Thanks go to three anonymous referees, one of whom graciously reviewed the article twice, which I greatly appreciate. The final form and some of the content is the result of their excellent work.
References and notes
- Williams, A.R., Beneficial mutations: real or imaginary—part II, J. Creation 28(2):75–82, 2014. Return to text.
- Fisher, R.A., The Genetical Theory of Natural Selection, Oxford University Press, p. 18, 1930. Return to text.
- Williams, A.R., Facilitated variation: a new paradigm emerges in biology, J. Creation 22(1):85–92, 2008. Return to text.
- Lightner, J.K., Designed to inhabit the earth: A review of The Plausibility of Life: Resolving Darwin’s Dilemma by Marc W. Kirschner and John C. Gerhart, Yale University Press, New Haven, CT, 2005, J. Creation 22(1):33–36, 2008. Return to text.
- Kirschner, M.W. and Gerhart, J.C., The Plausibility of Life: Resolving Darwin’s Dilemma, Yale University Press, New Haven, p. 227, 2005. Return to text.
- Pattee, H.H., The problem of biological hierarchy; in: Waddington, C.H. (Ed.), Towards a Theoretical Biology, vol. 3, Edinburgh University Press, pp. 117–136; p. 117, 1970. Return to text.
- Williams, A.R., Human genome decay and the origin of life, J. Creation 28(1):91–97, 2014. Return to text.
- Goldman, A., Ancient Life in the Information Age: What can bioinformatics and systems biology tell us about the ancestor of all living things?, The Scientist, 1 March 2014; the-scientist.com. Return to text.
- Research Spotlight: Jack Szostak, Making Life from Scratch, Origins of Life Initiative, origins.harvard.edu, accessed 14 March 2014. Return to text.
- Huxley, T.H., Biogenesis and Abiogenesis: Critiques and Addresses, Collected Essays, vol. vii, 1870, aleph0.clarku.edu, accessed 10 April 2014. Return to text.
- Britten, R.J. and Davidson, E.H., Gene regulation for higher cells: a theory, Science 165(3891):349–357, 1969. Return to text.
- The Davidson Laboratory, Research Summary Overview, its.caltech.edu, accessed 20 November 2013. Return to text.
- International prize for biology, jsps.go.jp, accessed 14 April 2014. Return to text.
- Davidson, E.H., Emerging properties of animal gene regulatory networks, Nature 468(7326):911–20, 2010. Return to text.
- Erwin, D.H. and Davidson, E.H., The evolution of hierarchical gene regulatory networks, Nature Reviews Genetics 10:141–148, 2009. Return to text.
- Davidson, E.H., The Regulatory Genome: Gene Regulatory Networks in Development and Evolutio, Academic Press, San Diego, CA, p. 195, 2006. Return to text.
- Davidson, E.H. and Erwin, D.H., An integrated view of precambrian eumetazoan evolution, Cold Spring Harbour Symposium on Quantitative Biology 74:65–80, 2009 | doi:10.1101/sqb.2009.74.042. Return to text.
- Parthenogenesis, wikipedia.org, accessed 14 March 2014. Return to text.
- Xu, Peng-Fei, X. et al., Construction of a vertebrate embryo from two opposing morphogen gradients, Science 344(6179):87–89, 2014. Return to text.
- Nusslein-Volhard, C., Coming to Life: How Genes Drive Development, Kales Press, Carlsbad, 2006. Return to text.
- Gardner, R.L., Can developmentally significant spatial patterning of the egg be discounted in mammals?, Human Reproduction Update 2(1):3–27, 1996. Return to text.
- Santos, F. et al., Active demethylation in mouse zygotes involves cytosine deamination and base excision repair, Epigenetics & Chromatin 6:39, 2013; epigeneticsandchromatin.com. Return to text.
- Li, L., Lu, X. and Dean, J., The maternal to zygotic transition in mammals, Molecular Aspects of Medicine 34(5):919–938, 2013 | doi: 10.1016/j.mam.2013.01.003. Return to text.
- Baroux, C. et al., The Maternal to Zygotic Transition in Animals and Plants, Cold Spring Harbour Symposium on Quantitative Biology 73:89–100, 2008; originally published online February 9, 2009. Return to text.
- Epigenetics, wikipedia.org, 6 May 2014. Return to text.
- Horvath, S., DNA methylation age of human tissues, Genome Biology 14:R115, 2013; genomebiology.com. Return to text.
- Tadros, W. and Lipshitz, H.D., The maternal-to-zygotic transition: a play in two acts, Development 136:3033–3042, 2009 | doi:10.1242/dev.033183. Return to text.
- Simonelig, M., Maternal-to-zygotic transition: soma versus germline, Genome Biology 13:145, 2012; genomebiology.com. Return to text.
- Siddiqui, N.U. et al., Genome-wide analysis of the maternal-to-zygotic transition in Drosophila primordial germ cells, Genome Biology 13:R11, 2012; genomebiology.com. Return to text.
- Genome expression, php.med.unsw.edu.au, accessed 11 April 2014. Return to text.
- Embryonic development, php.med.unsw.edu.au, accessed 3 April 2014. Return to text.





Readers’ comments
Comments are automatically closed 14 days after publication.