Journal of Creation 34(3):3–4, December 2020
Browse our latest digital issue Subscribe
Newly discovered jumping beetle mechanism inspires bionic design

A new catapult mechanism has recently been discovered in the hind legs of certain beetles, astounding researchers with its ability to pack a powerful punch. These ‘flea beetles’ have been observed to display incredible jumping abilities, surpassing that of fleas. A recent paper, published by the online peer-reviewed scientific journal ZooKeys, describes their ability:
“The jumping of flea beetles is an extremely effective method to avoid potential predators, as it allows beetles [to] quickly disappear from the leaf surface, where they spend most of their life. Blepharida sacra can jump up to 70 cm or 100 times more than its body length, while Longitarsus anchusae reaches a jump of 289 times its body length; the average acceleration of Psylliodes affinis during take-off can be up to 266 times the acceleration of gravity [g].”1
Scaled up to human size, that would be like a 6 ft (1.8 m) tall long-jump athlete leaping more than half a kilometre! When we consider that fighter pilots can sustain no more than 9g for a few seconds, it is remarkable how these beetles survive nearly 30 times that force. Furthermore, these bouncing bugs can perform more than 30 consecutive jumps without tiring.1
It took the collaborative efforts of a team of 13 scientists from America and China to discover the secret to the beetle’s bounce. They used a number of advanced techniques to visualise and measure the biological structures and power output of the insect’s jumping mechanism—micro-computed tomography (micro-CT scans), 3D computer reconstructions, high-speed filming, and light microscope dissection. The scientists measured the beetle’s jump, calculating its power and acceleration, and the figures derived are astounding! Regarding Psylliodes punctifrons, the authors report:
“The jump pushes individuals to a final velocity of 5.58 ± 0.5 m/s. The peak instantaneous power output (per unit mass) calculated for the hind legs in this species was 2.2 ± 0.1 × 105 W/kg, which is approximately 449 times that of the fastest-known muscle … and some 100–200 times that of a powerful rally car engine.”1
These startling statistics place these insects on the top podium, in terms of acceleration and power, of all known jumping insects, with second and third place going to fleas and leaf hoppers respectively.
A spring in its step
The mechanism responsible for the beetle’s jumping prowess turned out to be a sophisticated spring (figure 1).
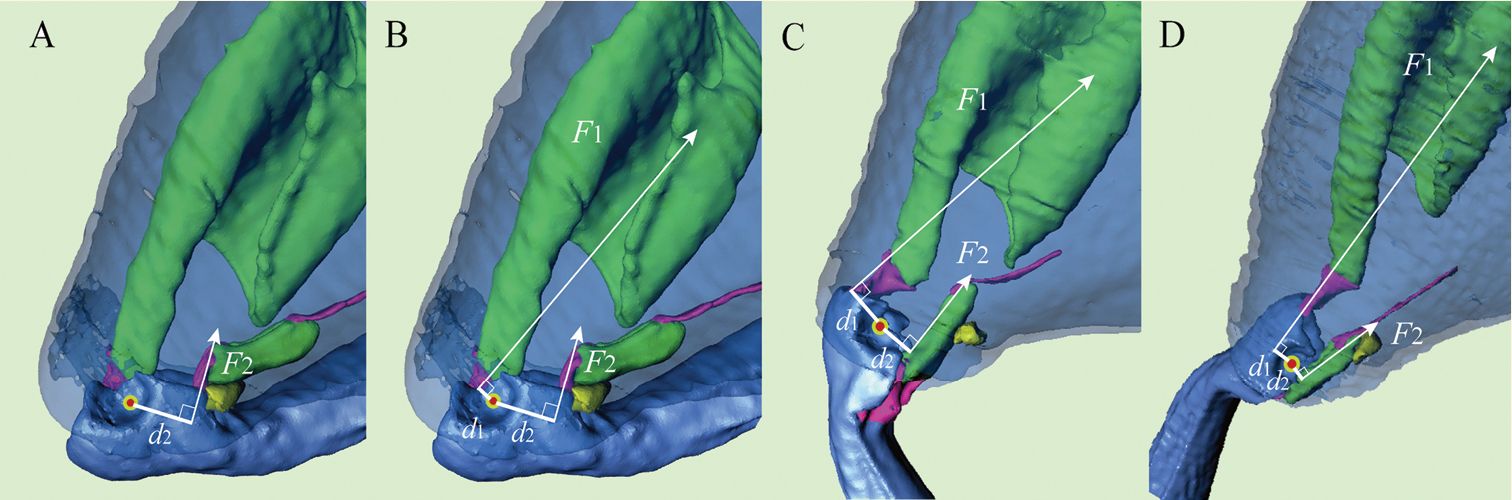
However, the authors of the paper fail to credit the Creator for the mechanism enabling these beetles’ dramatic locomotive abilities. Rather, they resort to empty platitudes that elevate evolution:
“ … nearly all the species of flea beetles have an ability to jump and this ability is commonly understood as one of the key adaptations responsible for its diversity.”1
Furthermore, a priori they resort to evolution to explain the specifics of the beetle mechanism: “Flea beetles have evolved an enormous independent spring to aid the storage of elastic potential energy.”1 But such an evolutionary explanation for the origin of the beetle’s jumping mechanism begs the question as to how they survived before they evolved this ability; the leap of logic for evolution being that beetles that fail to escape predators don’t leave progeny, so their genes are not passed on.
The jumping mechanism, situated in the beetle’s hind legs, is made of three moving parts (a latch, trigger, and spring) attached to muscles. The jump sequence has four phases, each of which increases the angle of the femur compared to the tibia. Phase I (crunching): muscles in the tibia contract, causing the tibia to flex. Phase II (co-contraction): extensor and flexor muscles in the tibia contract simultaneously, catching a triangular plate (see figure 1 ‘F2’) and inhibiting the tibia from extending further. Phase III (triggering and acceleration): the triangular plate is dislodged, causing the explosive release of stored energy. Phase IV (relaxation): the flea beetle is explosively launched upwards and the tibia muscles return to their initial relaxed position.
Good designs are typically repurposed for other applications. It has been discovered that a similar spring-catch-trigger mechanism exists in the snapping jaws of the Dracula ant2 and a four-bar snapping mechanism exists within the jaws of the snipefish (relatives of the sea-horses.)3 These kinds of apparatus give every appearance of being ‘irreducibly complex’ mechanisms. In other words, like Michael Behe’s mousetrap,4 all the component parts of the beetle’s jumping mechanism have to be present for the system to function. It cannot be further simplified without complete loss of function. The catapult mechanism in these beetles relies on the perfect balance of specifically shaped parts and their elastic properties. But how this mechanism could have evolved in an incremental way is impossible to conceive. The structure would need to be complete to afford the beetle a survival advantage to begin with.
Inspiration from creation

Inspired by the elegant jumping apparatus of flea beetles, the researchers proposed a robotic limb design (figure 2), using a similar mechanism. This section of their paper employs the term ‘design’ 12 times to describe the scientists’ robotic plan, itself based on their observations of the beetles. To produce such an engineering achievement, a great deal of planning and forethought must be applied—a far cry from the blind evolutionary processes that they propose for its origins. How much more brilliant a construction was conceived by the Divine Designer of the beetle, which the clever scientists tried to emulate?
Insect design
The spring apparatus of flea beetles is just one of many brilliant insect designs that have inspired engineers. Another example of an irreducibly complex jumping mechanism is the interlocking gear system discovered in nymph-stage leaf hoppers, the gears functioning as mechanical regulators for the insect’s jump.5 Gear systems, by their very nature, are irreducible, necessitating many integrated parts, each of specified complexity. These natural systems exhibit all the hallmarks of intelligent, engineering design. Conversely, these same structures are to be seen in the earliest fossils,6 demonstrating that there is no evidence of evolution.7 Evolution cannot account for what is observed in nature in terms of its information and complexity.8
Summing up the science
The flea beetle jumping mechanism discussed here can be added to an increasing list of ‘irreducibly complex mechanisms’ found in nature.9 Such systems are completely dependent upon each of their specific components for the mechanism to work correctly (or at all). Remove one component and the function of the whole is lost. This is axiomatic of human engineering systems, which require all their separate parts to function together. Intelligently created machines exhibit specified complexity, and require forethought and problem solving for their manufacture. This is as true for the robotic leg design as it is for the beetle design which inspired it.
References and notes
- Ruan, Y. et al., The jumping mechanism of flea beetles (Coleoptera, Chrysomelidae, Alticini), its application to bionics and preliminary design for a robotic jumping leg, ZooKeys 915:87–105, 24 February 2020 ǀ doi.org/10.3897/zookeys.915.38348. Return to text.
- Larabee F.J., A. Smith, A.A., Suarez, A.V., Snapjaw morphology is specialized for high-speed power amplification in the Dracula ant, Mystrium camillae, Royal Society B 5(12), July 2018 ǀ doi:10.1098/rsos.181447. Return to text.
- Longo, S.J., Goodearly T., Wainwright, P.C., Extremely fast feeding strikes are powered by elastic recoil in a seahorse relative, the snipefish, Macroramphosus scolopax, Proc. Roy. Society B 285(1882), 2018 ǀ doi.org/10.1098/rspb.2018.1078. Return to text.
- Wieland, C., The mousetrap man: Interview with Mike Behe, Creation 20(3):17, 1998. Return to text.
- Sarfati, J., Toothed gears in jumping insects, creation.com, 19 September 2013. Return to text.
- Cessna, S., Mighty mites stifle evolutionists, Creation 39(2):27, 2018. Return to text.
- Kruger, J., Fossilized insects show signs of stasis and rapid burial! J. Creation 28(1):8–9, 2014. Return to text.
- Gitt, W., Implications of the scientific laws of information—part 1, J. Creation 23(2):96–102, 2009, Implications of the scientific laws of information—part 2, J. Creation 23(2): 103–109, 2009. Return to text.
- Sarfati, J., Refuting Evolution 2, Master Books, AR, pp. 161–172, 2002. Return to text.





Readers’ comments
Comments are automatically closed 14 days after publication.