Journal of Creation 20(3):10–12, December 2006
Browse our latest digital issue Subscribe
Mitochondria—created to energize us
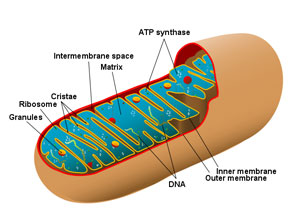
Mitochondria are small, membrane-bound organelles serving as energy generators in eukaryotic cells. Most cells have hundred to thousands of them, depending on their energy needs. Mitochondria are very good at what they do—they generate about 95% of a cell’s energy in the form of adenosine triphosphate (ATP) by oxidizing pyruvate (a by-product of anaerobic glycolysis) to CO2 and water. They are ovoid to filamentous in shape, generally ranging from one to seven micrometers in length (about the same size and shape as small bacteria). Since the discovery that mitochondria possess their own DNA, it has been frequently theorized that mitochondria evolved from ancient bacteria ingested by larger cells. This is known as the ‘endosymbiont theory’ of mitochondrial origin. Sometimes it is stated boldly:
‘More than a billion years ago, aerobic bacteria colonized primordial eukaryotic cells that lacked the ability to use oxygen metabolically. A symbiotic relationship developed and became permanent. The bacteria evolved into mitochondria, thus endowing the host cells with aerobic metabolism, a much more efficient way to produce energy than anaerobic glycolysis.’1
Sometimes it is stated more cautiously:
‘In the endosymbiont theory, the ancestor of the eukaryotic cell (we will call this organism a protoeukaryote) is presumed to have been a large, anaerobic, heterotrophic prokaryote that obtained its energy by a glycolytic pathway. Unlike present-day bacteria, this organism had the ability to take up particulate matter … . The endosymbiont theory postulates that a condition arose in which a large, particularly complex, anaerobic prokaryote took up a small aerobic prokaryote into its cytoplasm and retained it in a permanent state [emphasis added].’2
Whichever way it is stated, it is given an aura of authority and certainty by its frequent repetition in writings on cell biology. Many students find it convincing. However, like many evolutionary ideas, it may look solid from a distance, but gaps appear on close scrutiny.
The evidence for the endosymbiont theory revolves around selected similarities between mitochondria and bacteria, especially the DNA ring structure. However, these similarities do not prove evolutionary relationship. There is no clear pathway from any one kind of bacteria to mitochondria, although several types of bacteria share isolated points of similarity. Indeed, the scattered nature of these similarities has left plenty of room for a less-publicized ‘direct evolution’ theory of mitochondrial origin, in which they never had any free-living stage.3 There is enough diversity among the mitochondria of protozoa to make evolutionists wonder if endosymbiotic origin of mitochondria occurred more than once.4
Mitochondrial DNA
The endosymbiont theory implies that there should be considerable autonomy for mitochondria. This is not the case. Mitochondria are far from self-sufficient even in their DNA, which is their most autonomous feature. Mitochondria actually have most of their proteins coded by nuclear genes, including their DNA synthesis enzymes. For example, human mitochondria have 83 proteins, but only 13 are coded by mtDNA (mitochondrial DNA). Even those proteins which are coded by mtDNA often have large subunits that are coded by nuclear DNA. These nuclear-coded mitochondrial proteins must be labelled and transferred from the cytoplasm across two membranes. This intricate, hand-in-glove working between mtDNA and nuclear DNA presents a major difficulty for evolutionists. They have yet to propose a reasonable mechanism by which so many genes could be transferred intact (along with appropriate labelling and control mechanisms) to the nucleus.
Plants and other ‘lower creatures’ may have more mitochondrial genes than the higher animals do, but they still fall far short of the number necessary for free-living existence. Plants have also been found to have much more non-coding mtDNA than the ‘higher’ animals. Referred to as ‘junk DNA’ by evolutionists, it is held to have been eliminated by evolution from the mitochondrial genomes of the higher animals, to the point that humans have virtually no non-coding mtDNA. Evolution seems to be remarkably unpredictable in its handling of ‘junk DNA’, allowing it to accumulate ‘haphazardly’ in the nuclear DNA of higher animals and man, but ‘efficiently’ eliminating it from mtDNA. It doesn’t seem reasonable for evolutionists to have it both ways.
There are more important differences between mtDNA and nuclear or prokaryotic DNA. The main one is that the genetic code for mtDNA differs from the standard DNA code in slight but significant ways. Why? Evolutionists make much of the universality of the genetic code, saying that it offers strong support for common descent of all living things. If this is true—if the code is so highly conserved in evolution through over a billion years and millions of species—then even a few exceptions to the rule are hard to explain. (On the other hand, from a design standpoint the answer may lie in the simpler protein synthetic machinery served by mtDNA, which uses fewer tRNAs, and is less specific in codon recognition.) Lack of introns is another important difference. The ‘higher’ mtDNA has no introns, whereas nuclear DNA and some ‘lower’ mtDNA do have them. Again, the bacteria from which mitochondria are supposed to have evolved also lack introns. Thus, we’re asked to believe that the pre-mitochondrial bacteria sporadically evolved introns as they became ‘primitive’ mitochondria, and then lost them again as eukaryotic evolution ensued. As evolutionists grapple with the biochemical details, the endosymbiont theory becomes more and more cumbersome and vague.5
Intracellular control
As alluded to earlier, mitochondrial numbers are controlled within each cell by energy needs. They can also travel within cells on cytoskeletal microtubule ‘rails’ wherever energy is needed (near the ribosomes in pancreatic zymogen cells, near the proton pumps in gastric acid-secreting cells, etc.).6 This complex intracellular control is highlighted by a common pathological abnormality in which certain body cells become bloated by an oversupply of mitochondria. These cells, known to medicine as ‘oncocytes’, are packed by malformed or malfunctioning mitochondria, in which various mutations have been detected.7,8 Also, when mutated mitochondria derived from a maternal oocyte populate all of the body’s cells, the results can be devastating. A whole spectrum of degenerative multisystem diseases associated with mitochondrial mutations has been described recently, with more being discovered.9,10 Such diseases tend to affect tissues most heavily dependent on aerobic metabolism, such as neural and muscular tissue. These observable phenomena underscore the harsh reality that random changes in mitochondria or microbes do not produce complex new structures and regulatory systems, but rather disease and death.
It should also be pointed out that the engulfing of bacteria by larger cells is one of the commonest phenomena in nature, happening countless times each hour. Yet, nothing really like the formation of mitochondria has ever been observed. There may be rare modern examples of endosymbiosis between two different types of cells, such as the Chlorella algae within ‘green’ paramecia. Also, infecting or parasitic microbes can persist for a time inside of larger host cells due to encapsulation or other protective factors. Still, these events are far from the radical biotransformation demanded by the endosymbiont theory, and no one untainted by evolutionary preconceptions would ever dream of classifying mitochondria as once-separate life forms, as some evolutionists have suggested. It is essentially an ‘evolutionary miracle’, assumed to have happened in the past, but never seen or duplicated in the present.
Chloroplasts
Furthermore, if we accept this ‘naturalistic miracle’ of mitochondrial origin we are forced to conclude that the same miracle happened repeatedly. Evolutionists also postulate an endosymbiotic origin for chloroplasts, the organelles of photosynthesis in higher plants. Chloroplasts have their own DNA, once again with a ring structure. They are similar in some respects to present-day photosynthetic bacteria. However, because of biochemical variety among chloroplasts (like the mitochondria), evolutionists are once again forced toward the unlikely conclusion that their endosymbiotic origin occurred more than once!
‘According to this endosymbiont hypothesis, eucaryotic cells started out as anaerobic cells without mitochondria or chloroplasts and then established a stable endosymbiotic relationship with a bacterium, whose oxidative phosphorylation system they subverted to their own use … . Plant and algal chloroplasts seem to have been derived later from an endocytic event involving an oxygen-evolving photosynthetic bacterium. In order to explain the different pigments and properties found in the chloroplasts of present-day higher plants and algae, it is usually assumed that at least three different events of this kind occurred [emphasis added].’11
Although it is correctly admitted here that the endosymbiont scenario is actually only a hypothesis, it is presented as the only possibility. However, as shown above, the fine print admits that assumption and speculation are major components of this idea.
Why do mitochondria and chloroplasts have their own DNA? Evolutionists believe that it is a source of cellular inefficiency, and that evolution has been slowly phasing out cytoplasmic DNA over time. (This raises the obvious question of why there is any mtDNA left at all, to which the evolutionary response is that the process of elimination is either incomplete or arrested.) However, viewing mtDNA as inefficient may just be a reflection of our own ignorance of the fine details of mitochondrial function. Deeper knowledge may show that manufacture of certain mitochondrial protein subunits ‘on-site’ is very efficient, just as the energy-harnessing chemistry of the mitochondrial enzymes has been shown to be.
Conclusion
Given the enormous leaps of biochemical and genetic integration which are demanded by the endosymbiont theory, creationist skepticism is entirely justified. There is no compelling reason to believe it unless one has already decided that evolution is true. The creationist model, holding that structures may look similar because they were designed to do similar jobs, is a more reasonable way to view the miracle of mitochondria.
References
- DiMauro, S. and Schon, E., Mitochondrial respiratory-chain diseases, New England Journal of Medicine 358:2656, 2003. Return to text.
- Karp, G.,Cell Biology, 2nd edition, McGraw-Hill, New York, p. 773, 1984. Return to text.
- Karp, ref. 2, p. 775. Return to text.
- Alberts et al., Molecular Biology of the Cell, 3rd edition, Garland Publishing Inc., New York, p. 715, 1994. Return to text.
- Alberts et al., ref. 4, pp. 708, 709. Return to text.
- DiMauro et al., ref. 1, p. 2665. Return to text.
- Tallini, G., Oncocytic tumors, Virchow’s Archives 433:5, 1998. Return to text.
- Jih, D. and Morgan, M., Oncocytic metaplasia occurring in a spectrum of melanocytic nevi, American Journal of Dermatopathology 24(6):468, December 2002. Return to text.
- DiMauroet al., ref. 1, pp. 2656–2665. Return to text.
- Leonard, J. and Schapira, A., Mitochondrial respiratory chain disorders I: mitochondrial DNA defects, The Lancet 355:299–304, 2000. Return to text.
- Albertset al., ref. 4, pp. 714–715. Return to text.


Readers’ comments
Comments are automatically closed 14 days after publication.