Journal of Creation 18(2):28–36, August 2002
Browse our latest digital issue Subscribe
Why the Miller–Urey research argues against abiogenesis
Summary

Abiogenesis is the theory that under the proper conditions life can arise spontaneously from non-living molecules. One of the most widely cited studies used to support this conclusion is the famous Miller–Urey experiment. Surveys of textbooks find that the Miller–Urey study is the major (or only) research cited to prove abiogenesis. Although widely heralded for decades by the popular press as ‘proving’ that life originated on the early earth entirely under natural conditions, we now realize the experiment actually provided compelling evidence for the opposite conclusion. It is now recognized that this set of experiments has done more to show that abiogenesis on Earth is not possible than to indicate how it could be possible. This paper reviews some of the many problems with this research, which attempted to demonstrate a feasible method of abiogenesis on the early earth.
Contemporary research has failed to provide a viable explanation as to how abiogenesis could have occurred on Earth. The abiogenesis problem is now so serious that most evolutionists today tend to shun the entire field because they are ‘uneasy about stating in public that the origin of life is a mystery, even though behind closed doors they freely admit that they are baffled’ because ‘it opens the door to religious fundamentalists and their god-of-the-gaps pseudo-explanations’ and they worry that a ‘frank admission of ignorance will undermine funding’. 1
Abiogenesis was once commonly called ‘chemical evolution’, 2 but evolutionists today try to distance evolutionary theory from the origin of life. This is one reason that most evolutionary propagandists now call it ‘abiogenesis’. Chemical evolution is actually part of the ‘General Theory of Evolution’, defined by the evolutionist Kerkut as ‘the theory that all the living forms in the world have arisen from a single source which itself came from an inorganic form’. 3
Another reason exists to exaggerate abiogenesis claims—it is an area that is critical to proving evolutionary naturalism. 4 If abiogenesis is impossible, or extremely unlikely, then so is naturalism. 5 , 6 , 7 , 8
Darwin recognized how critical the abiogenesis problem was for his theory. He even conceded that all existing terrestrial life must have descended from some primitive life-form that was originally called into life ‘by the Creator’. 9 But to admit, as Darwin did, the possibility of one or a few creations is to open the door to the possibility of many others! If God made one type of life, He also could have made many thousands of different types. Darwin evidently regretted this concession later and also speculated that life could have originated in some ‘warm little pond’ on the ancient earth.
The ‘warm soup’ theory
Although seriously challenged in recent years, the warm soup hypothesis is still the most widely held abiogenesis theory among Darwinists. Developed most extensively by Russian atheist Alexandr Ivanovich Oparin (1894–1980) in his book, The Origin of Life, a worldwide best seller first published in 1924 (the latest edition was published in 1965). 10 Oparin ‘postulated that life may have evolved solely through random processes’ in what he termed a biochemical ‘soup’ that he believed once existed in the oceans. The theory held that life evolved when organic molecules that originally rained into the primitive oceans from the atmosphere were energized by forces such as lightning, ultraviolet light, meteorites, deep-sea hydrothermal vents, hot springs, volcanoes, earthquakes, or electric discharges from the sun. If only the correct mix of chemicals and energy were present, life would be produced spontaneously. Almost a half century of research and millions of dollars have been expended to prove this idea—so far with few positive results and much negative evidence. 11
What sequence?
Oparin concluded that cells evolved first, then enzymes and, last, genes. 12 Today, we recognize that genes require enzymes in order to function, but genes are necessary to produce enzymes. Neither genes nor cells can function without many complex structures such as ribosomes, polymerase, helicase, gyrase, single-strand–binding protein and scores of other proteins. Dyson concluded that Oparin’s theory was ‘generally accepted by biologists for half a century’ but that it ‘was popular not because there was any evidence to support it but rather because it seemed to be the only alternative to biblical creationism’. 13
The Miller–Urey research
Haldane, 14 Bernal, 15 Calvin 16 and Urey 17 all published research in an attempt to support this model—each with little, if any, success. Then, in 1953 came what some then felt was a critical breakthrough by Harold Urey (1893–1981) of the University of Chicago and his 23-year-old graduate student, Stanley Miller (1930–). Urey came to believe that the conclusion reached by ‘many’ origin-of-life researchers that the early atmosphere was oxidizing must have been wrong; he argued instead that it was the opposite, namely a reducing atmosphere with large amounts of methane. 18
Their ‘breakthrough’ resulted in front-page stories across the world that usually made the sensational claim that they had ‘accomplished the first step toward creating life in a test tube’. 19 Carl Sagan concluded, ‘The Miller–Urey experiment is now recognized as the single most significant step in convincing many scientists that life is likely to be abundant in the cosmos.’ 20 The experiment even marked the beginning of a new scientific field called ‘prebiotic’ chemistry. 21 It is now the most commonly cited evidence (and often the only evidence cited) for abiogenesis in science textbooks. 22

The Miller–Urey experiments involved filling a sealed glass apparatus with the gases that Oparin had speculated were necessary to form life—namely methane, ammonia and hydrogen (to mimic the conditions that they thought were in the early atmosphere) and water vapour (to simulate the ocean). Next, while a heating coil kept the water boiling, they struck the gases in the flask with a high-voltage (60,000 volts) tungsten spark-discharge device to simulate lightning. Below this was a water-cooled condenser that cooled and condensed the mixture, allowing it to fall into a water trap below. 23
Within a few days, the water and gas mix produced a pink stain on the sides of the flask trap. As the experiment progressed and the chemical products accumulated, the stain turned deep red, then turbid. 24 After a week, the researchers analyzed the substances in the U-shaped water trap used to collect the reaction products. 25 The primary substances in the gaseous phase were carbon monoxide (CO) and nitrogen (N 2 ). 21 The dominant solid material was an insoluble toxic carcinogenic mixture called ‘tar’ or ‘resin’, a common product in organic reactions, including burning tobacco. This tar was analyzed by the latest available chromatographic techniques, showing that a number of substances had been produced. No amino acids were detected during this first attempt, so Miller modified the experiment and tried again. 20 , 26
In time, trace amounts of several of the simplest biologically useful amino acids were formed—mostly glycine and alanine. 20 The yield of glycine was a mere 1.05%, of alanine only 0.75% and the next most common amino acid produced amounted to only 0.026% of the total—so small as to be largely insignificant. In Miller’s words, ‘The total yield was small for the energy expended.’ 27 The side group for glycine is a lone hydrogen and for alanine, a simple methyl (–CH 3 ) group. After hundreds of replications and modifications using techniques similar to those employed in the original Miller–Urey experiments, scientists were able to produce only small amounts of less than half of the 20 amino acids required for life. The rest require much more complex synthesis conditions.
Oxygen: enemy of chemical evolution
The researchers used an oxygen-free environment mainly because the earth’s putative primitive atmosphere was then ‘widely believed not to have contained in its early stage significant amounts of oxygen’. They believed this because ‘laboratory experiments show that chemical evolution, as accounted for by present models, would be largely inhibited by oxygen’. 28 Here is one of many examples of where their a priori belief in the ‘fact’ of chemical evolution is used as ‘proof’ of one of the premises, an anoxic atmosphere. Of course, estimates of the level of O 2 in the earth’s early atmosphere rely heavily on speculation. The fact is, ‘We still don’t know how an oxygen-rich atmosphere arose.’ 29
It was believed that the results were significant because some of the organic compounds produced were the building blocks of much more complex life units called proteins—the basic structure of all life. 30 Although widely heralded by the press as ‘proving’ that life could have originated on the early earth under natural conditions (i.e. without intelligence), we now realize the experiment actually provided compelling evidence for exactly the opposite conclusion. For example, without all 20 amino acids as a set, most known protein types cannot be produced, and this critical step in abiogenesis could never have occurred.
In addition, equal quantities of both right- and left-handed organic molecules (called a racemic mixture) were consistently produced by the Miller–Urey procedure. In life, nearly all amino acids that can be used in proteins must be left-handed, and almost all carbohydrates and polymers must be right-handed. The opposite types are not only useless but can also be toxic (even lethal) to life. 31 , 32
Was there a methane–ammonia atmosphere?
According to many researchers today, an even more serious problem is the fact that the atmosphere of the early earth was very different from what Miller assumed. ‘Research has since drawn Miller’s hypothetical atmosphere into question, causing many scientists to doubt the relevance of his findings.’ 33 The problem was stated as follows:
‘… the accepted picture of the earth’s early atmosphere has changed: It was probably O 2 -rich with some nitrogen, a less reactive mixture than Miller’s, or it might have been composed largely of carbon dioxide, which would greatly deter the development of organic compounds.’ 34
A major source of gases was believed to be volcanoes, and since modern-day volcanoes emit CO, CO 2 , N 2 and water vapour, it was considered likely that these gases were very abundant in the early atmosphere. In contrast, it is now believed that H 2 , CH 4 and NH 3 probably were not major components of the early atmosphere. Furthermore, many scientists now believe that the early atmosphere probably did not play a major role in the chemical reactions leading to life. 20
Although the composition of the atmosphere of the early earth is now believed to have consisted of large amounts of carbon dioxide, this conclusion still involves much speculation. Most researchers also now believe that some O 2 was present on the early earth because it contained much water vapour, and photodissociation of water in the upper layers of the atmosphere produces oxygen. 35 Another reason is that large amounts of oxidized materials exist in the Precambrian geological strata. 36
Yet another reason to conclude free oxygen existed on the early earth is that it is widely believed that photosynthetic organisms existed very soon after the earth had formed, something that is difficult for chemical evolutionary theories to explain. A 2004 paper argues from uranium geochemistry that there were oxidizing conditions, thus photosynthesis, at 3.7 Ga. 37 But according to uniformitarian dating, the earth was being bombarded by meteorites up to 3.8 Ga. So even granting evolutionary presuppositions, this latest research shows that life existed almost as soon as the earth was able to support it, not ‘billions and billions of years’ later. Even if the oxygen were produced by photodissociation of water vapour rather than photosynthesis, this would still be devastating for Miller-type proposals.
The dilution problem
Urey also speculated that the oceans in the ancient earth must have consisted of about a 10% solution of organic compounds that would be very favourable for life’s origin. 38 This level of organic matter would equal a concentration about 100 times higher than a modern American city’s sewer water. The total amount of extant organic compounds on the earth today could not produce even a fraction of that needed to achieve a concentration this high in the oceans.
Early hopes not realized
Modern replications of the Miller–Urey experiment using a wide variety of recipes, including low levels of O 2 , yield even lower amounts of organic compound than the original experiment. 39 To solve this problem, some researchers have speculated that small, isolated pools of water achieved the required level of concentration. The same problem remains: No feasible method exists to account for this source. Some even speculate that ‘submerged volcanoes and deep-sea vents—gaps in the earth’s crust where hot water and minerals gush into deep oceans—may have provided the initial chemical resources’. 40
To duplicate what might have happened in a primordial soup billions of years ago, scientists would need to mix the chemicals currently believed to be commonly found on the early earth, expose them to likely energy sources (usually speculated to be heat or radiation), and see what happens. No-one has performed this experiment, because we now know that it is impossible to obtain relevant biochemical compounds by this means. The Miller–Urey experiment held great hopes for the materialists, which have now given way to pessimism:
‘Soon after the Miller–Urey experiment, many scientists entertained the belief that the main obstacles in the problem of the origin of life would be overcome within the foreseeable future. But as the search in this young scientific field went on and diversified, it became more and more evident that the problem of the origin of life is far from trivial. Various fundamental problems facing workers in this search gradually emerged, and new questions came into focus … . Despite intensive research, most of these problems have remained unsolved.
‘Indeed, during the long history of the search into the origin of life, controversy is probably the most characteristic attribute of this interdisciplinary field. There is hardly a model or scenario or fashion in this discipline that is not controversial.’ 41
Some of these major problems will now be reviewed.
Functional proteins can exist only in very narrow conditions
To produce even non-functional amino acids and proteins, researchers must highly control the experiment in various ways because the very conditions hypothesized to create amino acids also rapidly destroy proteins. Examples include thermal denaturing of proteins by breaking apart their hydrogen bonds and disrupting the hydrophobic attraction between non-polar side groups. 42 Very few proteins remain biologically active above 50ºC, or below about 30ºC, and most require very narrow conditions. Cooking food is a good example of using heat to denature protein, and refrigeration of using cold to slow down biological activity. As any molecular biologist knows from daily lab work, the pH also must be strictly regulated. Too much acid or base adversely affects the hydrogen bonding between polar R groups and also disrupts the ionic bonds formed by the salt bridges in protein.
Cross-reactions
Miller had to deal with the fact that the common cross-reactions of biochemical reaction products cause destruction or interfere with amino acid production. All compounds that interfere with bonding must be isolated or they will destroy the proteins. Therefore, Miller had to remove many contaminants and impurities to obtain pure compounds that are not normally found in life. Otherwise, his apparatus would have produced many destructive cross-reactions.
This is no small problem. Many organic compounds, such as ethanol and isopropyl alcohol, function as disinfectants by forming their own hydrogen bonds with a protein and, as a result, disrupt the proteins’ hydrophobic interactions. 41 Alcohol swabs are used to clean wounds or to prepare skin for injections because the alcohol passes through cell walls and coagulates the proteins inside bacteria and other cells. Also, heavy metal ions such as Ag + , Pb 2+ and Hg 2+ must be isolated from proteins because they disrupt the protein’s disulfide bonds, causing the protein to denature. As an example, a dilute (1%) AgNO 3 solution is placed in the eyes of newborn babies to destroy the bacteria that cause gonorrhea. Many heavy metal ions are very toxic if ingested because they severely disrupt protein structure, especially enzymes.
Another problem is that many of the other compounds necessary for life, such as sugar, also react strongly with amino acids and affect amino acid synthesis. For example, Miller and others had to use a sugar-free environment in their experiments. 43 Miller stopped his experiment after just a few days, but if it had been allowed to go on, would the compounds he produced be destroyed or would they produce more complex amino acids? Research on Murchison meteorites found that natural conditions produce compounds much like Miller’s, and the result is stable—indicating that further time would not produce any new products. 44
The Miller–Urey experiments produced many other compounds aside from amino acids, resulting in a sticky mass that was actually further from the building blocks of life than were the postulated original precursor chemicals. Toxic compounds produced include cyanides, carbon monoxide, and others—actually most of the dark matter in the solution could not be identified by the researchers in 1953. 21
Undirected energy is disruptive
A critical question, ‘How much energy was necessary?’ has been much debated. 45 However, all forms of energy can disrupt protein, including all of those forms postulated to be important in abiogenesis, such as UV and lightning. 46
Many speculate that ultraviolet light was the source used to create life, but UV is highly toxic to life, and is, in fact, often used to destroy life (thus UV lights are used in hospitals to kill micro-organisms). The intensity of the destructive long wavelengths exceeds that of the constructive short ones, and the quantum efficiency of destruction is much higher than that for construction as well. 47 This means that destruction of amino acids is four to five orders of magnitude higher than construction.
In Miller’s UV experiments, he used a select wavelength to produce amino acids and screened out other wavelengths because they destroy amino acids. Yet both chemical-building and chemical-destroying light exists in sunlight. Amino acids are actually very delicate and readily break down under natural sunlight.
The Miller–Urey experiment also had strategically designed traps to remove the products from the radiation before they could be destroyed. On a primitive earth, any amino acids formed in the atmosphere would be destroyed long before they could be removed. Even the ocean would not protect them, because UV penetrates several metres of liquid water—you can even sunburn under water. This indicates that the conditions on the early earth could never have been favourable for abiogenesis.
Even simple movement can cause major protein damage: whipping cream or beating egg whites is one way of using mechanical agitation to deliberately denature protein (the whipping stretches the polypeptide chains until the bonds break).
Miller’s research has, for the reasons discussed above, helped us to better understand why life could not have emerged naturally. In a summary of the famous Miller–Urey origin-of-life experiment, Horgan concluded that Miller’s results at first seemed to
‘… provide stunning evidence that life could arise from what the British chemist J.B.S. Haldane had called the “primordial soup.” Pundits speculated that scientists, like Mary Shelley’s Dr. Frankenstein, would shortly conjure up living organisms in their laboratories and thereby demonstrate in detail how genesis unfolded. It hasn’t worked out that way. In fact, almost 40 years after his original experiment, Miller told me that solving the riddle of the origin of life had turned out to be more difficult than he or anyone else had envisioned.’ 48
Creating life in a test tube also turned out to be far more difficult than Miller expected. Scientists now know that the complexity of life is far greater than Miller (or anyone else) imagined in 1953, prior to the DNA revolution. 49 We now know that Miller’s
‘… much-touted experiments tell us very little about where real, functional proteins came from. Yet this inconvenient fact is rarely mentioned when headlines blare out the news that scientists have succeeded in creating the building blocks of life.’ 50
Life is far more complex than Miller believed
About the same time as Darwin, T.H. Huxley proposed a simple, two-step method of chemical recombination that he thought could explain the origin of the first living cell. Both Haeckel and Huxley thought that just as salt could be produced spontaneously by mixing powered sodium metal and heated chlorine gas, a living cell could be produced merely by mixing the few chemicals they believed were required. Haeckel taught that the physical basis of life is a substance he called ‘plasm’ of different types such as ‘colourless’ and ‘also red, orange, and other kinds of protoplasm’ that were comparable in complexity and texture to a pot of glue or cold jelly. 51
Haeckel also believed that the first single cell owed its ‘existence to spontaneous creation’ from inorganic compounds, primarily ‘carbon, hydrogen, oxygen, and nitrogen’. 52 Once the brew was mixed, Huxley concluded eons of time allowed spontaneous chemical reactions to produce the simple ‘protoplasmic substance’ that scientists once assumed was the essence of life. 53 As late as 1928, the cell was still thought to be relatively simple, and few scientists then questioned the belief that life commonly developed from relatively simple to relatively complex forms. They also thought evolution was ‘the formation of new structures and functions by combinations and transformations of the relatively simple structures and functions of the germ cells.’ 54
We now also realize, after a century of research, that the eukaryote protozoa, believed in Darwin’s day to be as simple as a bowl of gelatin, are actually enormously complex. A living eukaryotic cell contains many hundreds of thousands of different complex parts, including various motor proteins. These parts must be assembled correctly to produce a living cell, the most complex ‘machine’ in the universe—far more complex than a Cray supercomputer. Furthermore, molecular biology has demonstrated that the basic design of the cell is
‘… essentially the same in all living systems on earth from bacteria to mammals. … In terms of their basic biochemical design … no living system can be thought of as being primitive or ancestral with respect to any other system, nor is there the slightest empirical hint of an evolutionary sequence among all the incredibly diverse cells on earth.’ 55
This finding poses major difficulties for abiogenesis because life at the cellular level generally does not reveal a gradual increase in complexity as it allegedly ascends the evolutionary ladder from protozoa to humans. The reason why the molecular machinery and biochemistry of modern organisms is basically similar is that the basic biochemical requirements and constraints are the same for all life. 56
The polymerization problem
The Miller–Urey experiment left many critical questions unanswered, even such basic ones as, ‘How did the chemicals combine to form the first molecules of living organisms?’ 34 Chemicals do not produce life; only complex structures such as DNA and enzymes produce life. Also, even if the source of the amino acids and the many other compounds needed could be explained, how these many diverse elements became aggregated in the same area and then properly assembled themselves must still be dealt with. This problem is a major stumbling block to all abiogenesis theories because
‘… no one has ever satisfactorily explained how the widely distributed ingredients linked up into proteins. Presumed conditions of primordial earth would have driven the amino acids toward lonely isolation. That’s one of the strongest reasons that Wächtershäuser, Morowitz, and other hydrothermal vent theorists want to move the kitchen [that cooked life] to the ocean floor. If the process starts down deep at discrete vents, they say, it can build amino acids—and link them up—right there.’ 33
The amino acid assembly problem is complicated by the fact that amino acids are able to bond in many locations by many kinds of chemical bonds. To form polypeptide chains requires restricting the links to only peptide bonds, and only in the correct locations. All other bonds must be prevented from being formed, no easy task. In living cells, a complex control system involving enzymes exists to ensure that inappropriate bonds do not normally occur; without this system, these inappropriate bonds would destroy the proteins produced.

To form a protein, amino acids must link together to form a peptide bond, eliminating a water molecule. But there is a far greater tendency for the reverse to happen. This would be even more of a problem in water.
Another problem is that the strong thermodynamic tendency is for the peptide bonds to break down in water, not to form. 57 Without high-energy compounds such as ATP and enzymes, amino acids do not form the many polypeptides needed for life. Even dipeptides are difficult to form under natural conditions, yet the average protein is composed of around 400 amino acids.
Several recent discoveries have led some scientists to conclude that life may have arisen in submarine vents, where temperatures approach 350ºC. Unfortunately for both warm-pond and hydrothermal-vent theorists, the extreme heat has proven to be a major downfall of their theories. This is because high temperatures would accelerate the breakdown of amino acids, just as cooking meat breaks down the bonds, causing meat to become more tender. 57
Another theory is that abiogenesis may have been a consequence of the ‘self-ordering properties’ of biochemicals. 58 Just as electrostatic forces produce highly ordered crystals of salt from Na + and Cl – ions, so too, some Darwinists reasoned, in the same way, life may likewise self-assemble. This approach also has failed. For example, all nucleotide base pairs have an equal affinity to the sugar phosphate backbones on each side of the DNA molecule, and consequently, their order is not a result of bonding affinity differences but is due to information-directed assembly. In other words, the information does not derive from the DNA chemistry, but is instead external to it (see next section).
Miller himself has recognized that Kauffman’s research is not viable and, consequently, he was
‘… unimpressed with any of the current proposals on the origin of life, referring to them as “nonsense” or “paper chemistry.” He was so contemptuous of some hypotheses that, when I asked his opinion of them, he merely shook his head, sighed deeply, and snickered—as if overcome by the folly of humanity. Stuart Kauffman’s theory of autocatalysis fell into this category. “Running equations through a computer does not constitute an experiment,” Miller sniffed. Miller acknowledged that scientists may never know precisely where and when life emerged. “We’re trying to discuss a historical event, which is very different from the usual kind of science, and so criteria and methods are very different,” he remarked.’ 59
Information content
Another major reason the Miller–Urey experiments failed to support abiogenesis was that, although amino acids are the building blocks of life, a critical key to life is the information code stored in DNA (or, as in the case of retroviruses, RNA), depending on the sequence of nucleotides. This in turn provides the instructions for the amino acid sequences for the proteins, the machinery of life. 60 , 61 Michael Polanyi (1891–1976), former chairman of physical chemistry at the University of Manchester (UK) who turned to philosophy, affirmed a very important point—the information was something above the chemical properties of the building blocks:
‘As the arrangement of a printed page is extraneous to the chemistry of the printed page, so is the base sequence in a DNA molecule extraneous to the chemical forces at work in the DNA molecule. It is this physical indeterminacy of the sequence that produces the improbability of any particular sequence and thereby enables it to have a meaning—a meaning that has a mathematically determinate information content.’ 62
Paul Davies reinforced the point that obtaining the building blocks would not explain their arrangement:
‘… just as bricks alone don’t make a house, so it takes more than a random collection of amino acids to make life. Like house bricks, the building blocks of life have to be assembled in a very specific and exceedingly elaborate way before they have the desired function.’ 63
An analogy is written language. Natural objects in forms resembling the English alphabet (circles, straight lines, etc.) abound in nature, but this fact does not help to understand the origin of information (such as that in Shakespeare’s plays). The reason is that this task requires intelligence both to create the information (the play) and then to design and build the machinery required to translate that information into symbols (the written text). What must be explained is the source of the information in the text (the words and ideas), not the existence of circles and straight lines. Likewise, it is not enough to explain the origin of the amino acids, which correspond to the letters. Rather, even if they were produced readily, the source of the information that directs the assembly of the amino acids contained in the genome must be explained. 34
Another huge problem is that information is useless unless it can be read. But the decoding machinery is itself encoded on the DNA. The leading philosopher of science, Karl Popper (1902–1994), expressed the huge problem:
‘What makes the origin of life and of the genetic code a disturbing riddle is this: the genetic code is without any biological function unless it is translated; that is, unless it leads to the synthesis of the proteins whose structure is laid down by the code. But … the machinery by which the cell (at least the non-primitive cell, which is the only one we know) translates the code consists of at least fifty macromolecular components which are themselves coded in the DNA. Thus the code can not be translated except by using certain products of its translation. This constitutes a baffling circle; a really vicious circle, it seems, for any attempt to form a model or theory of the genesis of the genetic code.
‘Thus we may be faced with the possibility that the origin of life (like the origin of physics) becomes an impenetrable barrier to science, and a residue to all attempts to reduce biology to chemistry and physics.’ 64
That is, the genetic information and the required reading machinery form an irreducibly complex system. So far, it has eluded materialistic explanations. 65
The chirality problem
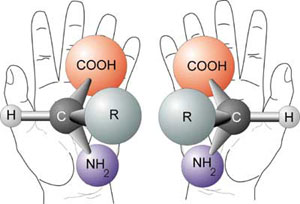
The two enantiomers of a generalized amino acid, where R is any functional group (except H)
What Sarfati 66 calls a ‘major hurdle’ is the origin of homochirality, the fact that all amino acid biomolecules with rare exceptions (such as some used in bacterial cell walls) are all left-handed; and with rare exceptions, all sugars, including those in nucleic acids, are right-handed. Those produced in a laboratory are a half left-handed and half right-handed mixture called a racemate. Even in the laboratory, chemists use pre-existing homochirality from a biological source in order to synthesize homochiral compounds. 60 Chiral molecules are dissymmetric—they exist as mirror images of each other, just as the right hand is a mirror image of the left hand (the word chiral comes from the Greek word for ‘hand’). The problem is left-handed sugars and right-handed amino acids can be toxic and prevent abiogenesis. Furthermore, most all enzymes are designed to work only with right-handed sugars and left-handed amino acids. All attempts to solve the chirality problem, including magnetochiral dichroism, have failed. 67
The legacy of the Miller experiment
A major unresolved question that ‘involves psychology and history more than chemistry’ is, ‘Why has the Miller–Urey experiment had such a strong impact on the origin-of-life field?’ 68 Shapiro concludes a major reason is that the experiment seems to imply that we are on the verge of understanding how life was created without intelligence or design. In the public mind (and in the minds of many scientists) this experiment psychologically supports abiogenesis. But the Miller–Urey results, and the many similar experiments completed since then, actually show the opposite of what the Miller–Urey experiment purported to demonstrate. Few textbooks actually analyze the results, and most uncritically accept this experiment as proof of how the building blocks of life were produced and then imply that the only task left was to determine how they were assembled.
My review of college textbooks found that most discussed the Miller–Urey experiments, some extensively, but few texts mentioned any of the problems. Most implied that the research has conclusively shown how the building blocks of life spontaneously generated. In part, due to the common claims in textbooks and museum exhibits, many people assume that a good, if not excellent, case exists for the Miller–Urey thesis. Davies noted that when he set out to write a book on the origin of life, he ‘was convinced that science was close to wrapping up the mystery of life’s origins’, but after spending ‘a year or two researching the field’, he is
‘… now of the opinion that there remains a huge gulf in our understanding … . This gulf in understanding is not merely ignorance about certain technical details, it is a major conceptual lacuna.’ 69
The Miller–Urey experiment is now an icon of evolution, presented in most all biology, zoology and evolution textbooks as clear evidence of abiogenesis, when it actually illustrates the many difficulties of chemical evolution. 22
The current status of the Miller–Urey line of research
In an interview with Stanley Miller, now considered one of ‘the most diligent and respected origin-of-life researchers’ in the world, after he completed his 1953 experiment, he ‘dedicated himself to the search for the secret of life’ but was also ‘quick to criticize what he feels is shoddy work’ in an effort to overcome the fact that the origin-of-life field has ‘a reputation as a fringe discipline, not worthy of serious pursuit’. 59 Miller vowed that one day
‘ … scientists would discover the self-replicating molecule that had triggered the great saga of evolution … . [and] the discovery of the first genetic material [will] legitimize Millers’ field. “It would take off like a rocket,” Miller muttered through clenched teeth. Would such a discovery be immediately self-apparent? Miller nodded. “It will be in the nature of something that will make you say, ‘… How could you have overlooked this for so long?’ And everybody will be totally convinced”.’ 59
This hope has become less realistic as our knowledge has advanced. What we have learned, especially during the past few years, makes it less likely than ever that abiogenesis was ever possible. 36 , 70 , 71 Yet the Miller–Urey experiment is now the classic, best-known origin-of-life experiment, cited in texts from high school to graduate school, in areas ranging from biology to geology and philosophy to religion. 20 , 22 Phillip Johnson summed up the whole Miller–Urey research problem as follows:
‘Because post-Darwinian biology has been dominated by materialist dogma, the biologists have had to pretend that organisms are a lot simpler than they are. Life itself must be merely chemistry. Assemble the right chemicals, and life emerges. DNA must likewise be a product of chemistry alone. As an exhibit in the New Mexico Museum of Natural History puts it, “volcanic gases plus lightning equal DNA equals LIFE!” When queried about this fable, the museum spokesman acknowledged that it was simplified but said it was basically true.’ 72
---------------------------------------------------
Conclusion
It is now recognized that the Miller–Urey line of research is simply a ‘revival of the antique notion of spontaneous generation’ because it
‘… suggests that given the primordial soup, with the right combination of amino acids and nucleic acids, and perchance a lightning bolt or two, life might in fact have begun “spontaneously”. The major difference is that according to what biologists customarily called spontaneous generation, life supposedly began this way all of the time. According to the “soup” suggestion, by contrast, it began this way only once in the immeasurably distant past.’ 73
We must conclude, as Ridley did, that the early forms of life, and how natural selection could shape them, are ‘so obscure at the primordial stage that we can only guess why complexity might have increased’.
Darwin thought about the question inconclusively. He once wrote to the geologist Charles Lyell about a question ‘which is very difficult to answer, viz. how at first start of life, when there were only simplest organisms, how did any complication of organisms profit them? I can only answer that we have not facts enough to guide any speculation on the subject.’ We have more facts now, but they are still inadequate, and Darwin’s answer still holds. 74
When confronted with this evidence, supporters of abiogenesis argue that science must be naturalistic, and we have no choice but to tell the best story we have, even if it is not a complete or even accurate story. 4 Although widely heralded by the popular press for decades as ‘proof’ that life originated on the early earth entirely by natural conditions, the Miller–Urey experiments have actually provided compelling evidence for exactly the opposite conclusion. This set of experiments—more than almost any other carried out by modern science—has done much more to show that abiogenesis is not possible on Earth than to indicate how it could be possible.
Acknowledgments
I want to thank Tim Wallace, Bert Thompson, Wayne Frair, Clifford Lillo and John Woodmorappe for their comments on an earlier draft of this article.
References and notes
- Davies, P., The Fifth Miracle: The Search for the Origin and Meaning of Life , Simon & Schuster, New York, pp. 17–18, 1999. Return to text.
- Dickerson, R.E., Chemical evolution and the origin of life, Scientific American 239 (3):62–102, 1978. Return to text.
- Kerkut, G.A., Implications of Evolution , Pergamon, Oxford, UK, p. 157, 1960. Return to text.
- Johnson, P., Reason in the Balance; The Case Against Naturalism in Science, Law and Education , InterVarsity Press, Downers Grove, 1995. Return to text.
- Dover, G., Looping the evolutionary loop; review of: The Origins Of Life: From The Birth Of Life To The Origin Of Language , Nature 399 :217–218, 1999. Return to text.
- Dawkins, R., Climbing Mount Improbable , W.W. Norton, New York, 1996. Return to text.
- de Duve, C., Vital Dust: Life as a Cosmic Imperative , Basic Books, New York, 1995. Return to text.
- Denton, M., Nature’s Destiny; How the Laws of Biology Reveal Purpose in the Universe , The Free Press, New York, 1998. Return to text.
- Darwin, C., Origin of Species , reprint of 6th edition, P.F. Collier, New York, p. 316, 1900. This concession to theism was absent in earlier editions. Return to text.
- Oparin, A., The Origin of Life , New York, Dover, 1965. Return to text.
- Poundstone, W., Carl Sagan; A Life in the Cosmos , Henry Holt, New York, 1999. Return to text.
- Dyson, F., Origins of Life , Cambridge University Press, p. 31, 1990. Return to text.
- The Origin of Life, users.rcn.com/jkimball.ma.ultranet, accessed 13 May 2004. Return to text.
- Bernal, J.D., The physical basis of life, Physical Society of London Proceedings, Section A 62 :537, 1947. Return to text.
- Haldane, J.B.S., Rationalist Annual , 1928; reprinted in: Science and Human Life , Harper and Brothers, New York, 1933. Return to text.
- Calvin, M., Reduction of carbon dioxide in aqueous solutions by ionizing radiation, Science 114 :416–418, 1951. Return to text.
- Urey, H., The Planets: Their Origin and Development , Yale University Press, New Haven, pp. 149–157, 1952. Return to text.
- Lewis, R., Life , 3rd edition, WCB McGraw-Hill, Boston, p. 153, 1999. Return to text.
- Colson, C. and Pearcey, N., How Now Shall We Live? Tyndale House, Wheaton, p. 69, 1999. Return to text.
- Shapiro, R., Origins; A Skeptics Guide to the Creation of Life on earth , Summit Books, New York, p. 99, 1986.
- Lahav, N., Biogenesis: Theories of Life’s Origin , Oxford University, New York, 1999. Return to text.
- Wells, J., Icons of Evolution , Regnery, Washington, 2000. Return to text.
- Campbell, N.A., Mitchell, L.G. and Reece, J.B., Biology Concepts and Connections , 3rd edition, Addison Wesley Longman, Inc., San Francisco, 2000. Return to text.
- Miller, S.L., A production of amino acids under possible primitive earth conditions, Science 117 :528–529; p. 528, 1953. Return to text.
- Shapiro, ref. 20, p. 100. Return to text.
- Miller, S.L., Production of some organic compounds under possible primitive earth conditions, J. American Chemical Society 77 :2351–2361, 1955. Return to text.
- Miller, ref. 24, p. 529. Return to text.
- Fox, S. and Dose, K., Molecular Evolution and the Origin of Life , Marcel Dekker, New York, p. 44, 1977. Return to text.
- Sleep, N.H., Biogeochemistry; oxygenating the atmosphere, Nature 410 :317–319; 2001, p. 319. Return to text.
- Horgan, J., The End of Science: Facing the Limits of Knowledge in the Twilight of the Scientific Age , Addison-Wesley, Reading, p. 130, 1996. Return to text.
- Jamali, F., Lovlin, R., Corrigan, B.W., Davies, N.M. and Aberg, G., Stereospecific pharmacokinetics and toxicodynamics of ketorolac after oral administration of the racemate and optically pure enantiomers to the rat, Chirality 11 (3):201–205, 1999. Return to text.
- Coppedge, J.F., Probability of left-handed molecules, CRSQ 8 :163–174, 1971. Return to text.
- Simpson, S., Life’s first scalding steps, Science News 155 (2):24–26, 1999; p. 26. Return to text.
- Flowers, C., A Science Odyssey: 100 Years of Discovery , William Morrow and Company, New York, p. 173, 1998. Return to text.
- Scherer, S., Could life have arisen in the primitive atmosphere? J. Molecular Evolution 22 (1):91–94, 1985; p. 92. Return to text.
- Thaxton, C., Bradley, W. and Olsen, R., The Mystery of Life’s Origin; Reassessing Current Theories , ch. 5, Philosophical Library, New York, 1984. Return to text.
- Rosing, M.T. and Frei, R., U-rich Archaean sea-floor sediments from Greenland—indications of >3700 Ma oxygenic photosynthesis, Earth and Planetary Science Letters 217: 237–244, 2004. Return to text.
- Urey, ref. 17, p. 153. Return to text.
- Campbell, N.A., Biology , Benjamin/Cummings, Redwood City, 1993. Return to text.
- Campbell et al. , ref. 23, p. 321. Return to text.
- Lahav, ref. 21, p. 50. Return to text.
- Timberlake, K., Chemistry: An Introduction to General, Organic, and Biological Chemistry , Benjamin Cummins, San Francisco, 2001. Return to text.
- Witham, L., By Design: Science and the Search for God , Encounter Books, San Francisco, p. 106, 2003. Return to text.
- Witham, ref. 43, p. 98. Return to text.
- Vogel, G., RNA study suggests cool cradle of life, Science 283 (5399):155–156, 1999. Return to text.
- Williams, E.L., The evolution of complex organic compounds from simpler chemical compounds: is it thermodynamically and kinetically possible? CRSQ 4 :30–35, 1967. Return to text.
- Hulett, H.R., Limitations on Prebiological Synthesis, Journal of Theoretical Biology 24 :56–72, 1969. Return to text.
- Horgan ref. 30, p. 138. Return to text.
- Yockey, H.P., Information Theory and Molecular Biology , Cambridge University Press, Cambridge, p. 336, 1992. Return to text.
- Colson and Pearcey, ref. 19, p. 71. Return to text.
- Haeckel, E., The Last Link: Our Present Knowledge of the Descent of Man , Adam and Charles Black, London, p. 120, 1898. Return to text.
- Haeckel, ref. 51, p. 48. Return to text.
- Meyer, S., DNA and other designs, First Things , April, pp. 30–38, 2000; p. 31. Return to text.
- Conklin, E.G., Embryology and evolution; in: Mason, F. (Ed.), Creation by Evolution , Macmillan, New York, pp. 63–64, 1928. Return to text.
- Denton, M., Evolution: A Theory in Crisis , Adler and Adler, Bethesda, p. 250, 1986. Return to text.
- Cairns-Smith, A.G., The first organisms, Scientific American 252 (6):90–100, 1985. Return to text.
- Sarfati, J. , Origin of life: the polymerization problem , J. Creation 12 (3):281–284, 1998. Return to text.
- Kauffman, S., The Origins of Order , Oxford University Press, New York, 1993; At Home in the Universe , Oxford University Press, New York, 1995. Return to text.
- Horgan, ref. 30, p. 139. Return to text.
- Pigliucci, M., Where do we come from? A humbling look at the biology of life’s origin, Skeptical Inquirer 23 (5):21–27, 1999. Return to text.
- Dembski, W.A., The Design Inference: Eliminating Chance Through Small Probabilities , Cambridge University Press, Cambridge, England, 1998. Return to text.
- Polanyi, M., Life’s irreducible structure, Science 160: 1308, 1968. Return to text.
- Davies, P., Life force, New Scientist 163 (2204):27–30, 1999; p. 28. Return to text.
- Popper, K.R., Scientific reduction and the essential incompleteness of all science; in: Ayala, F. and Dobzhansky, T. (Eds.), Studies in the Philosophy of Biology , University of California Press, Berkeley, p. 270, 1974. Return to text.
- Sarfati, J., Self-replicating Enzymes? A critique of some current evolutionary origin-of-life models , J. Creation 11 (1):4–6, 1997. Return to text.
- Sarfati, J., Origin of life: the chirality problem , J. Creation 12 (3)263–266, 1998. Return to text.
- Sarfati, J., Origin of life and the homochirality problem: is magnetochiral dichroism the solution? , J. Creation 14 (3)9–12, 2000. Return to text.
- Shapiro, ref. 20, p. 107. Return to text.
- Davies, ref. 1, p. 17. Return to text.
- Levy, M. and Miller, S.L, The stability of the RNA bases: Implications for the origin of life, Proc. Nat. Acad. Sci. USA 95 :7933–7938, 1998. Return to text.
- Behe, M., Darwin’s Black Box , Basic Books, New York, 1996. Return to text.
- Johnson, P., Weekly Wedge Update , April 30, p. 1, 2001. Return to text.
- Serafini, A., The Epic History of Biology , Plenum, New York, p. 292, 1993. Return to text.
- Ridley, M., The Cooperative Gene; How Mendel’s Demon Explains the Evolution of Complex Beings , The Free Press, New York, p. 84, 2001. Return to text.





















Readers’ comments
Comments are automatically closed 14 days after publication.