Supercapacitor electrode design inspired by leaves on branches

A capacitor is an electrical component that stores energy as opposite charges on two parallel plates. The greater the area of the plates, and the smaller the distance between them, the greater the capacitance (ability to store charge). Supercapacitors achieve their very high capacitance because the separation between the plates is less than a nanometre (one thousand-millionth of a metre).
Supercapacitors don’t yet have the storage capacity of the best batteries. But they can be charged and discharged much more quickly, and retain their ability even after millions of charge/discharge cycles. So they are very good for the regenerative braking used in hybrid cars, for example.
Now, some researchers from the UCLA Henry Samueli School of Engineering and Applied Science and four other universities have made far more powerful supercapacitors.1 They did so by greatly increasing the electrode surface area—inspired by tree design.2 Because of the arrangement of the branches and leaves, the leaves have a very large surface area to absorb the carbon dioxide required for photosynthesis.
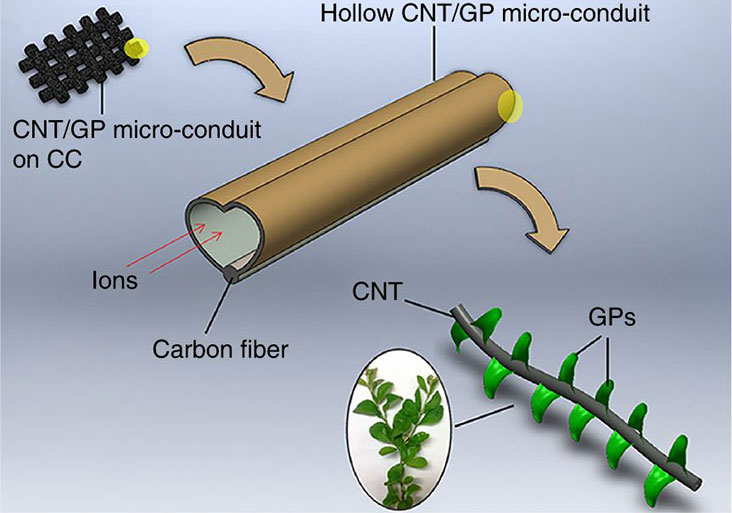

For the electrodes, they used two relatively new special structures made of carbon. For the ‘branches’: carbon nanotubes, hollow tubes only 20 to 30 nanometres in diameter; and for the ‘leaves’: graphene, a sheet of a single layer of carbon atoms in a hexagonal lattice, which also stabilized the electrode. Their supercapacitors had 30% more capacitance than other carbon-based capacitors of the same mass, and 30 times the capacitance for a given area. And thanks to the large area for exchanging charges, their design produced 10 times more power. It also retained 95% of its capacity after over 10,000 charging cycles, and performed well in both acidic and high-temperature environments.
Team leader Tim Fisher, professor of mechanical and aerospace engineering at UCLA, said:
We often find inspiration in nature, and plants have discovered the best way to absorb chemicals such as carbon dioxide from their environment. In this case, we used that idea but at a much, much smaller scale—about one-millionth the size, in fact.1
These engineers made a brilliant design, but by their own admission, their design was a mere copy. So how much more credit should be given to the Designer of the original,3 who made the green plants on Day 3 of Creation Week, a day before He made the sun?
References and notes
- UCLA Henry Samueli School of Engineering of Applied Science, Inspired by nature: Design for new electrode could boost supercapacitors’ performance, phys.org, 26 February 2018. Return to text.
- Guoping Xiong and six others, Bioinspired leaves-on-branchlet hybrid carbon nanostructure for supercapacitors, Nature Communications 9:790, 2018 | doi:10.1038/s41467-018-03112-3. Return to text.
- See many more examples at ‘Scientists copying nature (biomimetics)’, creation.com/biomimetics. Return to text.





Readers’ comments
Comments are automatically closed 14 days after publication.