Carbon-14 dating—explained in everyday terms
[This article of mine, written in 1979, is obviously outdated and should not be relied upon. It is retained as part of our archive on Creation magazine, but for a current summary of CMI’s view on carbon-14 dating, the reader is referred to Chapter 4, What about carbon dating? of CMI’s The Creation Answers Book. —CW.]
Summary
An attempt to explain this very important method of dating and the way in which, when fully understood, it supports a ‘short’ timescale. In fact, the whole method is a giant ‘clock’ which seems to put a very young upper limit on the age of the atmosphere. The article is in straightforward language and the non-technical reader could profitably work through it.
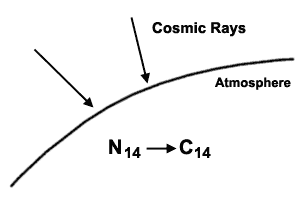
Carbon-14 (14C) or radiocarbon as it is often called, is a substance manufactured in the upper atmosphere by the action of cosmic rays. Ordinary nitrogen (14N) is converted into 14C as shown to the right. Ordinary carbon is carbon-12 (12C). We find it in carbon dioxide in the air we breathe (CO2), which of course is cycled by plants and animals throughout nature, so that your body, or the leaf of a tree, or even a piece of wooden furniture, contains carbon. When 14C has been formed, it behaves just like ordinary carbon (12C), combining with oxygen to give carbon dioxide (14CO2), and also gets freely cycled through the cells of all plants and animals. The difference is this: once 14C has been formed, it begins to decay radioactively back to 14N, at a rate of change which can be measured. If we take a sample of air, and measure how many 12C atoms there are for every 14C atom, this is called the 14C/12C ratio. Because 14C is so well ‘mixed up’ with the 12C, we find that this ratio is the same if we sample a leaf from a tree, or a part of your body. Think of it like a teaspoon of cocoa mixed into a cake dough—after a while, the ‘ratio’ of cocoa to flour particles would be roughly the same no matter which part of the cake you sampled. The fact that the 14C atoms are changing back to 14N doesn’t matter in a living thing—because it is constantly exchanging carbon with its surroundings, the ‘mixture’ will be the same as in the atmosphere and in all living things.
As soon as it dies, however, the 14C atoms which decay are no longer replaced by new ones from outside, so the amount of 14C in that living thing gets smaller and smaller as time goes on. Another way of saying it is that the 14C/12C ratio gets smaller. In other words, we have a ‘clock’ which starts ticking at the moment something dies.
Obviously this only works for things which once contained carbon—it can’t be used to date rocks and minerals, for example. We know how quickly 14C decays, and so it becomes possible to measure how long it has been since the plant or animal died.
But wait—how do we know what the 14C/12C ratio was to start with? We obviously need to know this to be able to work out at what point the ‘clock’ began to tick. We’ve seen that it would have been the same as in the atmosphere at the time the specimen died. So how do we know what that was? Do scientists assume that it was the same as it is now? Well, not exactly. It is well known that the industrial revolution, with its burning of huge masses of coal, etc. has upset the natural carbon balance by releasing huge quantities of 12C into the air, for example. Tree-ring studies can tell us what the 14C/12C ratio was like before the industrial revolution, and all radiocarbon dating is made with this in mind. How do we know what the ratio was before then, though, say thousands of years ago? It is assumed that the ratio has been constant for a very long time before the industrial revolution. Is this assumption correct? (For on it hangs the whole validity of the system.) Why did W.F. Libby, the brilliant discoverer of this system, assume this?
Libby knew that 14C was entering and leaving the atmosphere (and hence the carbon cycle). Because Libby believed that the Earth was millions of years old, he assumed that there had been plenty of time for the system to be in equilibrium. This means that he thought that 14C was entering the atmosphere as fast as it was leaving—calculations show that this should take place in about 30,000 years, and of course the Earth was much older than that, said the geologists. Imagine a tank with water flowing in at a certain rate, and flowing out again at the same rate (see diagram below). This system is in equilibrium. If you saw it for the first time, you wouldn’t be able to work out how old it was—how long it had been since it was ‘switched on’.
| A = B, therefore system is in equilibrium |  |
 |
If A is greater than B, then the tank is ‘filling up’ |
Was Libby right in this assumption? Was the 14C entering and leaving the system at the same rate? In his day, the measurements and calculations, which he knew about, showed that 14C was entering the system some 12–20% faster than it was leaving. Imagine the same tank, this time it is not yet full and the top tap is flowing more quickly than the bottom one is leaking out—this gives you a way of measuring how long ago the whole system was ‘switched on’ and it also tells you that that can’t have been too long ago (see diagram above).
Libby knew that if these figures were correct, it would mean that the atmosphere was young, so he dismissed the results as being due to experimental error! (We are not implying dishonesty here, merely showing how powerfully the evolutionary/uniformitarian concepts of Earth history influence great scientists to mould or discard evidence which appears to contradict that viewpoint.) What about modern measurements, using advanced technology such as satellites? Unfortunately for the ‘old-Earth’ advocates, the studies of such renowned atmospheric physicists as Suess and Lingenfelter show that 14C is entering the system some 30–32% faster than it is leaving it. The model of radiocarbon dating which Libby developed, using his incorrect ‘uniform’ assumption, must therefore be corrected to fit the facts about 14C—let us call the new, corrected model the ‘non-uniform’ model. What does this mean? It implies that if the 14C is still ‘building up’, we can calculate how old the whole system is—this puts an upper limit on the age of the atmosphere of some 7 to 10,000 years. Also, it means that a thousand years ago, the 14C/12C ratio in the atmosphere was less than today (because the 14C was still building up). Therefore a specimen which died a thousand years ago will show an older age than its true age. Two thousand years ago, specimens would have still less 14C to start with, so they have an even greater error. In other words, the further you go back, the more you have to shrink the radiocarbon dates to make them fit the facts. Remember that this correction is based on measurable scientific data, not on any creationist preconceptions.
We need to consider two other effects:
- If, as many creationists propose, there was a vast water vapour canopy around the Earth before the Flood, then this would have shielded the atmosphere from much of the cosmic radiation. Therefore, the amount of 14C in the pre-flood world would have been very small, perhaps even negligible. So a specimen from before the flood would appear to be ‘very old’ or even on ‘infinite’ age because it had so little 14C in it, making it look as if it had been decaying for tens of thousands of years. [Editor’ note: See Noah’ Flood: Where did the water come from? for updated information on the ‘canopy theory’.]
- The measured exponential decay of the Earth’s magnetic field, as described by Dr Thomas Barnes (see article last issue) suggests that as you go back in history, the strength of the field increases rapidly. A stronger magnetic field would mean more protection against cosmic rays, therefore again much less 14C produced and again this give artificially ‘old’ ages the more you go back in time.
In summary then:
- The 14C in the atmosphere is not in equilibrium, but is building up rapidly.
- This seems to put an upper limit on the age of the whole system in the order of 10,000 years.
- All radiocarbon dates have to be adjusted from the obviously incorrect ‘uniform’ model which is still in use today, and when this is done there is a shrinking in these dates. The older the date, the greater the reduction.
- The protective water vapour canopy and the greater magnetic field before the flood would decrease 14C levels in the past, causing greatly exaggerated 14C ‘ages.’
In any case, even the incorrect ‘uniform’ model has given, in many cases, serious embarrassment to evolutionists by giving ages which are much younger than those he expects in terms of his model of earth history. Consider this—if a specimen is older than 50,000 years, it has been calculated, it would have such a small amount of 14C that for practical purposes it would show an ‘infinite’ radiocarbon age. So it was expected that most deposits such as coal, gas, petrified trees, etc. would be un-dateable by this method. In fact, of 15,000 dates in the journal Radiocarbon to 1968, only three were classed ‘un-dateable’—most were of the sort which should have been in this category. This is especially remarkable with samples of coal and gas supposedly produced in the carboniferous 100 million years ago! Some examples of dates which contradict orthodox (evolutionary) views:
- Coal from Russia from the ‘Pennsylvanian’, supposedly 330 million years old, dated at 1,680 years. [Incorrect—this particular sample was charcoal from Kyrgyzstan, not coal from Russia.1 But numerous instances of carbon-14 in coal have been reliably recorded—see Refs2,3 below, as well as chapter on Carbon Dating from our Creation Answers Book. Ed.]
- Natural gas from Alabama and Mississippi (Cretaceous and Eocene, respectively)—should have been 50 to 135 million years old. 14C gave dates of 30,000 and 34,000, respectively.
- Bones of a sabretooth tiger from the LaBrea tar pits, supposedly 100,000 years old, gave a date of 28,000.
- A block of wood from the Cretaceous (supposedly more than 70 million years old) found encased in a block of Cambrian rock (hundreds of millions of years earlier), gave a date of 4,000 years.
Remember that all these dates are using the incorrect ‘uniform’ model. (refs. in The World and Time, pub. by Creation-Science Research Center)
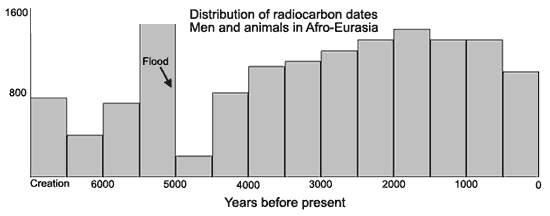
A question which could be asked after all this is: does radio-carbon, adjusted to fit the ‘non-uniform’ model, give any independent evidence of a worldwide catastrophe such as the Flood? Certainly if there was such a Flood, as we maintain from several other lines of evidence and reasoning, most living things would have perished, and so we would expect a ‘cut-off’ point at this time. In other words, going into the past, we should reach a period of time in which there is a sharp reduction in the number of specimens compared to the period just older than that, and as we went forward in time, we would expect a gradual buildup, as plant and animal populations recovered their numbers.
Such a study has been done by Dr Robert Whitelaw. Using the 15,000 published dates previously mentioned after adjusting them as described, he grouped them into 500 year ‘blocks’ and found a dramatic drop-off about 5,000 years ago, with a worldwide distribution (Speak to the Earth, Ed. G. Howe. Presb. & Ref. Pub. Co, p. 331). Readers are referred to this article for other interesting conclusions about these dates. [Editor’s note: The graph below was reproduced from a sketch in the original magazine. Note that the data presented does not necessarily endorse a particular age for the Earth, but reveals a pattern consistent with a recent creation and global flood model.]
We see, then, that far from being an embarrassment to the creationist who believes in a young Earth, the radiocarbon method of dating—when fully understood in accordance with modern atmospheric data—gives powerful support to his position.
References
- Vinogradov, Alexander Pavlovich et al., Radiocarbon dating in the Vernadsky Institute I–IV, Radiocarbon 8(1):292–323, 1966; p. 319. Return to text.
- Giem, Paul, Carbon-14 content of fossil carbon, Origins 51:6–30, 2001. Return to text.
- Lowe, D.C., Problems associated with the use of coal as a source of 14C-free background material, Radiocarbon 31(2):117–120, 1989. Return to text.



Readers’ comments
Comments are automatically closed 14 days after publication.