An amazing desert trio
Adapted to the heat

After the global upheaval of the Genesis Flood, the Earth’s climate changed dramatically. Even after the Flood waters had receded, the oceans were still warm, causing substantial evaporation and precipitation. There was much volcanic dust, etc. in the air, reflecting sunlight and so cooling the land. All this led to the Ice Age.
After a few centuries, the warmer oceans cooled, resulting in considerably less precipitation. Previously well-watered regions of the Earth became dry. Several parts of the world became deserts—such as the Sahara, which still shows evidence of its previous lush climate.1
To survive there, animal populations would have needed to adapt to these extremely hot and dry environments, and some clearly have. God did not create each of these well-adapted species separately. Many can still hybridize with similar species that are not desert-adapted, showing they are descended from the same Genesis kind.
That does not mean that the animals on the Ark had to display features ready to allow them to live in desert areas. Rather, creatures in general were created with lots of genetic variation, and for some kinds this would have included information for features that would be advantageous in very dry conditions. Natural selection in a desert environment would then eliminate creatures lacking this information, or at least its full expression, and thus increase the frequency of this information in the remaining population.
Adaptation to desert environments
One feature that is advantageous in a desert environment is small size. Smaller animals give off heat more easily than larger ones, and don’t need to eat or drink as much. Another advantageous feature is slower metabolism. This can be done by something known as torpor. This is a temporary state of slower metabolism like hibernation, accompanied by lower body temperature. A third, easier way to literally escape the heat is simply to be active at night (nocturnal lifestyle) or at dawn/dusk (crepuscular lifestyle), and sleep during the day.
Table 1. Several similarities and unique traits between the sand cat, fennec, and mulgara.
| Trait | Sand cat | Fennec | Mulgara |
| Active time | At dusk, night | At night | At night |
| Body size | Smallest wild cat species | Smallest fox species | Similar to mouse |
| Burrows | Present | Present | Present |
| Colouration | Light brown to yellow | Light reddish brown to yellow | Brown |
| Diet | Rodents, insects, snakes | Rodents, insects, birds | Rodents, insects, reptiles |
| Ears | Large for prey detection, heat loss | Large for prey detection, heat loss | Average size |
| Distribution | Sahara, Middle East, Central Asia | Sahara, Arabian Peninsula | Australian deserts and grasslands |
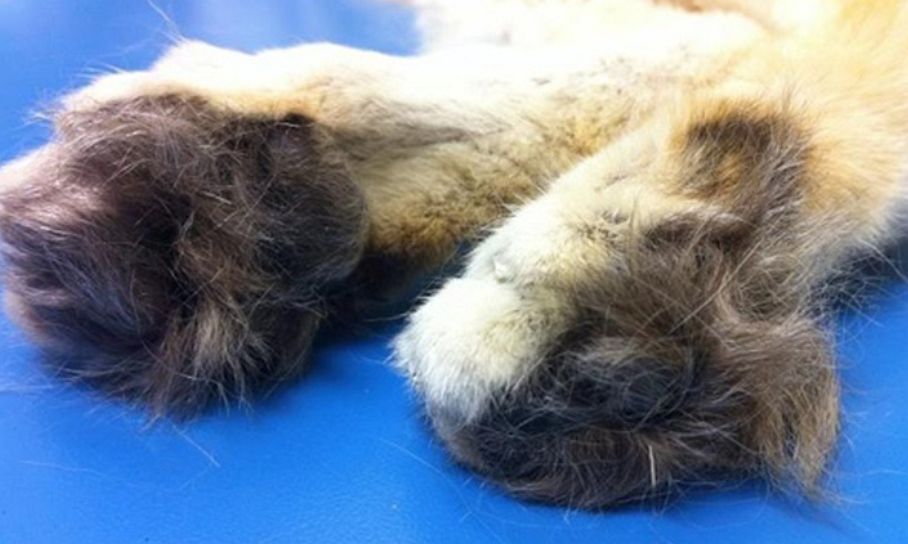
| Paws | Fur on paws and between toes | Furry paws | No fur on paws |
| Torpor | Unknown | Unknown | Present |
| Urine excretion | Unknown | Highly concentrated | Highly concentrated |
It’s not hard to see how the potential for considerable variation in body size, for example, would have been present in many, perhaps most, of the animal kinds coming off the Ark. So, if a population of animals enters a desert, individuals with genes for larger body size would more readily overheat and die before reproducing.

Individuals with genes for smaller size would cope better, and their offspring would inherit those ‘small-size’ genes (see ‘How natural selection depletes information’ on p. 31). The same would be true in principle for a number of other ways for animals to ‘beat the heat’, though it does not mean that every single kind of animal on the Ark was ‘front-loaded’ with genes for adapting to future desert conditions.
Let’s now look at a trio of fascinating animals well-adapted to desert environments.
The specialized sand cat
The sand cat (Felis margarita, shown on p. 29) is the only cat to live mainly in desert areas, far from water. The geographic range of the sand cat extends from the Sahara Desert to parts of the Middle East and Central Asia, but some live in captivity. Sand cats are one of the smallest species of cat in the wild. When the temperature gets too high in the day, they retreat to their burrows and are active only at dusk or at night.2
Sand cats have large, triangular ears (which also help to dissipate heat), and a wide external ear canal, which gives them a fine sense of hearing, up to five times better than other cats.3 This is needed to find prey including small rodents, insects, and even venomous snakes in underground burrows. Though they may drink water when near human habitation, they mostly get whatever water they need to survive from their food. Water is a byproduct from ‘burning’ of food, besides being present in the food itself.
Another interesting sand cat trait is the presence of pads of fur on its paws and between its toes to protect it from the scorching desert sand (shown on p. 30). Sand cats also have a light brown-yellow coat of fur which helps them camouflage themselves in their desert environment.
The phantom fennec fox
If the sand cat isn’t exotic enough, it’s as if this creature had a twin brother, the fennec fox (Vulpes zerda, above right). The fennec, the world’s smallest fox species, overlaps geographically with the sand cat. It survives the desert heat in similar ways and has a similar diet.

It is light reddish brown to yellow and has large ears which give off heat and enable it to detect prey underneath the sand. Fennecs’ ears can be as long as 40% of their own body length. As if these adaptive similarities were not enough, the fennec also has furry paws used for protection against the hot sand.
To release more heat, the fennec’s breathing rate can rise from a mere 23 to an amazing 690 breaths per minute, thirty times faster!
Fennecs eat insects, rodents, lizards, birds, eggs, roots, fruits, and leaves. Like the sand cat, the fennec gets whatever water it needs from its food. The fennec also excretes highly concentrated urine in order to retain water.
Just like sand cats, fennecs are also active during the night when they hunt for their prey. During the day, they hide in their burrows to keep cool.4
The maverick mulgara

The last animal in our desert trio is the mulgara (below, also p. 28 left), found in the arid deserts and grasslands of Australia. It is a mouse-like marsupial—i.e. like kangaroos and koalas, the mother raises her young in a pouch on her belly. Its diet is made up of insects, reptiles, and small rodents, which it hunts at night. Like the fennec, the mulgara excretes very concentrated urine to retain water, since it rarely drinks. The two mulgara species are Dasycercus cristicauda, the crest-tailed mulgara, and the smaller brush-tailed mulgara (Dasycerus blythi).
The frequency and length of torpor varies in mulgaras based on their diet. Individuals which mainly eat vertebrates have shorter and fewer bouts of torpor than those which eat invertebrates, which have a lower energy content.5
Can evolution explain it?
Natural selection, as we’ve seen, is a real phenomenon that aids adaptation to an environment. This is likely how e.g. today’s many different cat species—lions as well as sand cats—arose from one pair of their kind post-Flood, rapidly adapting to different ecological niches. But it can only ‘choose’ from genetic information that is present. It generates no new genes, instead it eliminates them.
Such examples of adaptation generally mean that the genetic information on which selection could act was already present on the Ark—even if not expressed or brought to the fore in the population.
Evolution, however, claims that all features in any animal arose by mutations (copying errors in the genes) over millions of years, on which selection could then act. But the likelihood of a random genetic change creating true design information is extremely small; observations confirm that mutations are, overall, degenerative.
How natural selection depletes information
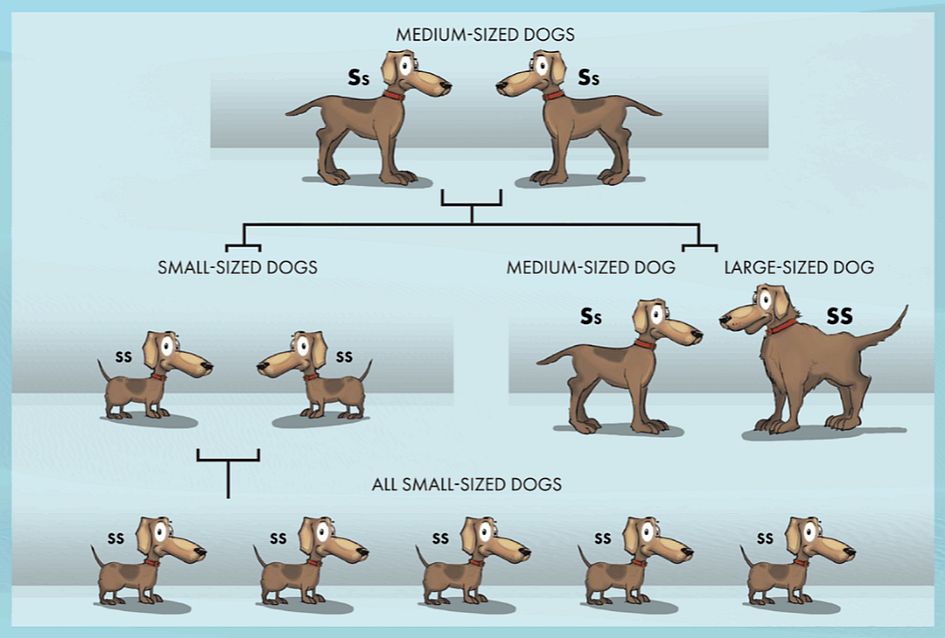
In this simplified example using dogs, a single gene pair is shown under each dog as coming in two possible forms. One form of the gene (S) carries instructions for large size, the other (s) for small size.
In row 1, we start with medium-sized animals (Ss) interbreeding. Each of the offspring can get one of either gene from each parent to make up their two genes.
In row 2, we see that the resultant offspring can have either large (SS), medium (Ss) or small (ss) size. If in a desert environment, e.g., only the small-sized dogs survive, they will be the only ones to pass their genes on to the next generation (row 3). From then on, all the dogs will be a new, small variety. Note that these are better adapted to their environment, but no new genes have been added. In fact, genes have been lost from the population, i.e. there has been a loss of genetic information, the opposite of what microbe-to-man evolution needs in order to be credible.
So the population is more specialised but less able to adapt to future environmental changes (e.g. back to large size). Not only genes for larger size were lost, but potentially several other genes the larger animals carried if not represented in the surviving animals. Genes on their own are not selected; it’s the whole creature and all the genes they carried.
At best, mutations could only explain trivial examples of adaptive change. In almost all cases, they are ‘breaking’ something, not ‘making’ something. For example, a mutation that stunts an animal’s growth could be an advantage in the desert, but it is still breaking the growth mechanism, not creating anything new.
Change within preset limits
A better explanation is that the information needed for the adaptive features in these three animals was already present in the cat and dog kind, and the mulgara’s kind,6 at creation and after the Flood. All cats obviously have genes for fur; these are in every cell of the body, but control mechanisms ensure they are only expressed where needed. A deleterious mutation in that mechanism, giving rise to fur on paws, would normally be selected against, but here would be an advantage, and thus selected for.
The genetic potential for a range of ear sizes was already there too; the animals with larger ears would have the advantage in desert conditions. Such change is not unlimited, however, since it is constrained by the information in the gene pool of the kind as a whole.
Evolution also has difficulty explaining why these three animals adapt to the heat in such very similar ways. See the list in Table 1 for comparison. For example, the sand cat and the fennec are both small, both have large ears and furry paws, and both have similar colouration and are both nocturnal. These exact same combinations of adaptations are unlikely to have happened just by chance, as if two painters each independently painted a picture of the same objects without conferring beforehand.
Conclusion
In summary, this amazing desert trio is no friend of evolutionary theory. Their ability to cope with extreme heat shows the marvellous handiwork and forethought of our Almighty Creator.
References and notes
- Oard, M., The problem of the wet Sahara, J Creation 31(1):3–4, 2017. Return to text.
- The sand cat, nationalzoo.si.edu/animals/sand-cat, accessed 7 Nov 2019. Return to text.
- Huang, G.T. and 3 others, Mammalian ear specializations in arid habitats: structural and functional evidence from sand cat (Felis margarita), J. Comp. Physiol. A: Neuroethol. Sens. Neural Behav. Physiol. 188(9):663–81, 2002. Return to text.
- The fennec fox; nationalzoo.si.edu/animals/fennec-fox, accessed 4 Nov 2019. Return to text.
- Pavey, C.R. and 3 others, Vertebrate diet decreases winter torpor use in a desert marsupial, Naturwissenschaften 96(6):679–83, 2009. Return to text.
- Possibly incorporating quolls and Tasmanian devils. Return to text.




Readers’ comments
Comments are automatically closed 14 days after publication.