Do koalas prove that humans got part of their DNA from viruses?
Or are they parts of the genome which cause diversity?
Does some of our DNA really come from viruses?

Some evolutionists allege that 8% of the human genome originated from viruses.1 This number is ambiguous, since different authors include different genetic elements as viruses, such as SINEs and LINEs. For example, Alu elements make up at least 11% of the human genome.2 According to another study, 22.4% of the genome is covered by endogenous retroviruses (ERVs).3 They claim that this came about when viruses infected humans and inserted their DNA into the human genome. This process is believed to have taken place over millions of years of evolutionary time. However, this has never been scientifically proven by direct observation.
Researchers from the University of Massachusetts Medical School have been studying the spread of a koala retrovirus (KoRV) as it infects the genomes of koalas from northern to southern Australia. Based on this, these researchers claim that a similar process could have also happened with other viruses in our own genome in the distant evolutionary past.
But how do retroviruses such as KoRV manage to insert themselves in the genome of a species? A retrovirus is a special kind of virus that inserts its own genome into the DNA of a host organism. Hence the ‘retro’ in retrovirus. A well-known example of a retrovirus is HIV, which infects our immune cells. Usually, retroviruses spread horizontally, from individual to individual, sometimes causing illness as they go along. But this process does not allow them to insert themselves permanently as part of the genetic material of a given species. What researchers want to see is whether the virus can transmit itself vertically, from parent to offspring. This would mean that the virus has become a permanent part of the genome of the species and will persist in the species in the long run.
During horizontal transfer, viruses spread between the body cells of two individuals. But during vertical transfer, viruses jump from a somatic cell (a regular body cell) to the germline. The germline is made up of the reproductive cells, sperm and eggs. If viral DNA is incorporated into the genome of the cells that give rise to the sperm or egg cells, then the virus will survive to spread in following generations.
What do viruses allegedly do once inside the genome?
Those viruses which have become totally fixed within the genome after a long time are called endogenous retroviruses (ERVs). According to evolutionary theory, in this manner, the genome would be much like a molecular fossil graveyard. In this graveyard we would find the remains of different kinds of retroviruses, as a sort of genetic ‘fossil record’ of infections by retroviruses over millions of years. The older the retrovirus, the more copies it has in the host genome, since it infected the genome much longer ago—or so the story goes.4 This is how evolutionists try to explain the presence of 30–40 different families of ‘viruses’ present in 1–1,000 copies each, allegedly introduced over the past 30 million years (according to the evolutionary timescale) in the human genome.
Once inside the human genome, several things can happen to retroviruses. Usually they can lose their function and degenerate into functionless DNA. They can also cause illnesses, such as cancer. This happens when the retrovirus disrupts a gene by inserting itself right in the middle of it. This would be like inserting a long paragraph in Russian into the middle of an English novel. The result would be unreadable. Evolutionists amazingly claim that retroviruses can also be re-programmed or repurposed to take part in cellular processes. One frequently cited example is the syncytin gene, which allegedly originated from a retrovirus, and which was re-coded to take part in the fusion of certain cells to form part of the placenta.5
How does the koala retrovirus spread?
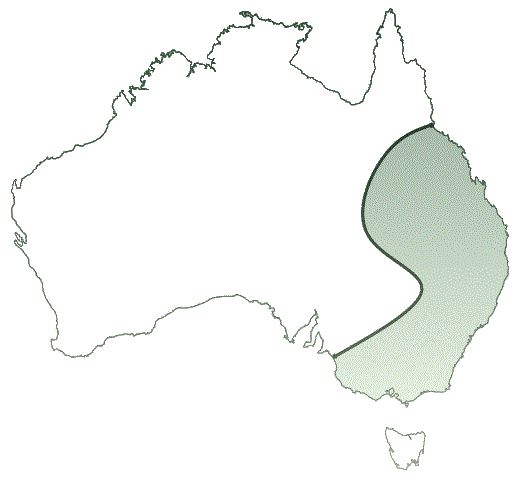
The KoRV has been spreading over the past 100 years in Australia, according to evolutionists. In the north, koalas all have copies of the KoRV in their genome, whereas a small number in the south do not (see fig. 1).6 It is interesting to note that KoRV causes lymphomas7 in infected koalas. Besides this, KoRV suppresses the koalas’ immune system, making them susceptible to infectious diseases, such as chlamydiosis.8
Contrary to evolutionary theory, the previously described process is one major roadblock for viruses to integrate into the host genome. If the presence of the KoRV in the koala genome is so advantageous to koalas, then they shouldn’t be falling victim to natural selection by such debilitating illnesses such as cancer.
But there is yet a second major obstacle for the virus to fully integrate into the host genome. Like the cellular immune system, the genome has a way of dealing with these disruptive retroviral invaders. The immune system can distinguish its own cells from that of viruses, bacteria or other invaders. This kind of immune response is also present in the genome at a molecular level. On a genetic level the genome is capable of distinguishing between its own genes and the genes of viruses (such as KoRV) and bacteria. This is possible, since the structure of virus genes is different from those of koala.

The genes of eukaryotes such as human or koala are more complex than those of bacteria or viruses. Eukaryotic genes (as in koalas and humans) are made up of several units (called exons, which are interspersed with segments of DNA called introns) which can be cut and pasted in different combinations (i.e. spliced) into a final mRNA product to specify a protein.9 On the other hand, the genes of bacteria and viruses are made up of one single unit, and are not spliced (see fig. 2).
The KoRV genome is made up of three genes, gag, pol and env, the last of which codes for the protein envelope of the virus (see fig. 3). Of these, the gag and pol genes are prokaryotic, and are not spliced as the eukaryotic genes, whereas the env gene is. To deactivate the gag and pol genes of KoRV, the koala cell produces so-called piRNA molecules10. The piRNA sequence is a complement of the gag and pol genes. The gag and pol mRNA and the piRNA are all single stranded, meaning they form a single helix. When piRNA binds to either the gag or pol gene, they then form a double helix. In this way the viral RNA is deactivated (fig. 4).11 Interestingly, piRNA is enriched in host tissues such as the testis, right where the retrovirus needs to insert itself into the germline.
Does viral invasion of host genomes make sense?
The evolutionary picture of viruses invading the genomes of animal species and slowly integrating as functional units into their host’s genome over millions of years runs into numerous problems. First, The spread of KoRV within koalas has been happening for only 100 years, not millions of years. This timeframe better fits the biblical timeline rather than the evolutionary timescale.
Second, if KoRV is useful in the long run, then why does it cause such debilitating diseases such as cancer? The researchers studying the KoRV have even suggested producing a vaccine against the retrovirus to save the koalas from extinction.1
How is it that so many inactive copies of KoRV survive in the koala genome? Around 8% or more of the human genome is made of retroviruses. If the KoRV is retained in the koala genome as junk DNA, it would most likely have been removed from the genome. It would cost the cell much too much energy and material to keep the junk DNA intact for an indefinite length of time. This would be like printing a book with extra pages of gibberish at the end over and over again. It would be superfluous to keep junk DNA in the genome for such a long period of time without eliminating it. It is much more likely that ERVs such as KoRV already had some function in the genome to begin with.

Also, it is a wide stretch of the imagination to say that retroviruses can contribute to a new function within the genome. If viral DNA is inserted at random into the genome, it is much more likely to disrupt existing genes. A blindfolded painter will most likely mess up the painting he is working on by just applying random brushstrokes to it.
Lastly, why is it that host piRNA already exists in the genome with a sequence which is complementary to that of the gag and pol genes? It is as if the host genome had been able to look ahead and had already come up with the piRNA sequence in order to counteract and deactivate the gag and pol genes of the invading retrovirus. But evolution is built upon blind chance and cannot know ahead of time what kind of challenges will face the organism. This is like stumbling upon a tribe of isolated forest dwellers, one of whom can speak perfect English with you. It is more likely that piRNA already existed within the host genome as a design element to keep the levels of the KoRV in check.
Why creation is a better explanation
It is possible to reinterpret retroviruses according to creation theory. Accordingly, retroviruses could have originated from within the host genome instead of invading it from outside. This had to have been the case in any scenario, since viruses are dependent upon host cells. This means that the cell’s genome had to predate viruses. Viruses are incapable of surviving without a host cell. That is, the ERVs are more likely rogue products of the genomes of their hosts. And the parts of genomes that look like ERVs are then seen as designed to do what they do (and well).
Research will increasingly reveal the functions of these misnamed ‘retrovirus elements’. Dr Ian Macreadie, at the time researching the retrovirus HIV, suggested this way back in 1999.12 Since viruses take up such a large portion of the genome, they must have some sort of function. Viruses are capable of transporting genes besides their own between bacteria, for example.
According to the variation-inducing genetic element, or ‘VIGE’ hypothesis, certain genes in host genomes do not originate from retroviruses from outside.13 Rather these genes are influenced by transposable genetic elements,14 which can alter the regulation of genes. When these transposable elements leave copies of themselves behind, they ensure that the new variation that they brought about can be inherited.15,16

The transposable elements which cause the variation within the host genome are the gag and pol genes. The gene that they regulate is the env protein. The syncytin gene is attached to the env gene of the so-called ERV-W element which is expressed in a part of the placenta called the syncytiotrophoblast. Yet another regulatory transposable element called the trophoblast specific enhancer (TSE) is responsible for expressing the syncytin gene only in the placenta and nowhere else. Syncytin is essential for successful pregnancy, so how did mammals reproduce before the putative ERV-W infected the mammal line and then fortuitously developed this function? It is all rather fanciful wishful thinking on behalf of evolutionists.
Conclusion
Evolutionists claim that a large portion of the human genome is littered with remains of viral infections, with remains of virus DNA littered here and there. This has never been observed experimentally, so researchers must look elsewhere to see if this process really does take place in nature.
Evolutionists claim that retroviruses such as the koalavirus are invading the genome of their host species. They say that this way these viruses can help the evolution of the host genome by inserting novel DNA into it. This idea is rather implausible. It goes against the logic of natural selection, because the koalas become sick due to viral infection. Therefore, those koalas infected with the virus would be eliminated, together with any copies of the virus with them.
But there is a second hurdle at the cellular level the koalavirus must jump before it can fully get integrated into the genome of its host. This is a molecular selection system, which eliminates genes which the cell does not recognize as its own. This involves special RNA molecules which bind to the virus genes, thereby removing them from cellular traffic and making it impossible for them to become a permanent part of the host genome.
Certain mobile retrovirus particles originally could have moved from site to site within the genome, thereby changing the way genes are regulated within the host genome. It makes much more sense if retroviruses were originally designed as part of the host genome and escaped from the cell after the accumulation of a mutations.
References and notes
- Joseph, A., What a Koala Virus Tells Us about the Human Genome, scientificamerican.com, 1 Nov 2019. Return to text.
- Deininger, P., Alu elements: know the SINEs, Genome Biology 12(12):236, 2011. Return to text.
- Conley, A., Jordan, K., Piriyapongsa, J., Retroviral promoters in the human genome, Bioinformatics 24(14):1563–7, 2008. Return to text.
- Stoye, J.P., Koala retrovirus: a genome invasion in real time, Genome Biology 7(11):241, 2006. Return to text.
- Dupressoir, A., Vernochet, C., Bawa, O., et al., Syncytin-A knockout mice demonstrate the critical role in placentation of a fusogenic, endogenous retrovirus-derived, envelope gene, Proc Natl Acad Sci U S A 106(29):12127–32, 2009. Return to text.
- Tarlinton, R., Meers, J., Young, P., Biology and evolution of the endogenous koala retrovirus. Cell Mol Life Sci. 65(21):3413–21, 2008. Return to text.
- A lymphoma is one of several types of cancer affecting the immune system. Return to text.
- Chlamydia is a genus of bacteria. Return to text.
- The mRNA is a copy of a gene and is called messenger RNA. Its sequence can be translated into a sequence of amino acids that make up a protein. Return to text.
- Technically, piRNA stands for Piwi-interacting RNAs. This is a short RNA molecule (18–34 letters long), which is formed from a region in the genome which doesn’t code for genes. Return to text.
- Yu, T., Koppetsch, B.S., Pagliarani, S., et al., The piRNA Response to Retroviral Invasion of the Koala Genome, Cell 179(3):632–643, 2019. Return to text.
- Creation in the research lab. Return to text.
- Terborg, P., The ‘VIGE-first hypothesis’—how easy it is to swap cause and effect, Journal of Creation 27(3):105–112, 2013. Return to text.
- Transposable genetic elements can move themselves around in the genome from one location to the other. Return to text.
- Terborg, P., The design of life: part 4—variation inducing genetic elements and their function, Journal of Creation 23(1):107–114, 2009. Return to text.
- Doyle, S., Large scale function for ‘endogenous retroviruses’, J. Creation 22(3):16, 2008. Return to text.



Readers’ comments
Comments are automatically closed 14 days after publication.