Journal of Creation 26(3):29–34, December 2012
Browse our latest digital issue Subscribe
A theistic paleontologist with dubious theology and little-better science
A review of Evolution and Belief: Confessions of a Religious Paleontologist by Robert J. Asher
Cambridge University Press, New York, 2012

This evolutionary book differs from most others in that it goes deeper, presenting claims seldom found elsewhere. While unambiguously hostile to them, it generally avoids making straw men of creationists.
God and evolution?
Asher professes belief in God. He repeats hoary antibiblical myths, such as the one about it teaching that stars hang from a solid firmament (p. 22).1 He derides certain Christian social teachings, and makes an implied equation of the Islamic terrorists of 911 with Christian fundamentalists (p. 130). Apart from being false and offensive, they are irrelevant to the theme of his work, and only detract from it.
He frowns upon what he calls an ‘intervening’ God, and One who is ‘outside the process’. He cites Psalms 147:9 on God providentially feeding the ravens. There are a number of fallacies in his reasoning. To begin with, if Genesis 1 is not factual and authoritative to Asher, then why suddenly is Psalm 147:9? Second, we are not in an either/or situation as Asher imagines. The fact that God feeds His creatures providentially does not nullify His feeding them miraculously (e.g. John 6:11–14). Third, Asher is confusing origins science and operations science, blurring them together according to his tacit evolutionary preconceptions. The fact that God operates providentially (operations science: Thomas Aquinas’ cited example of God providing heat through a fire) itself tells us nothing about how He had acted when He created the universe (origins science). Asher provides a lucid example of his confusion as he compares belief or non-belief in God, for the origins of the universe, with belief or non-belief in Thomas Edison when it comes to understanding how the light bulb works. Edison is irrelevant to the functioning of the light bulb (operations science) but is crucial to the design and creation of the light bulb (origins science).
Not surprisingly, Asher tries to ‘attach’ God to evolution by citing the ‘who’ and ‘why’ behind it. In actuality, evolutionary theories emphasize purposelessness, thus making ‘who’ or ‘why’ effectively meaningless. Asher also rejects the Anthropic Principle, that the universal constants must have values consistent with the conditions for known life (p. 204).
The author also tries to leave room for God by saying that evolution is not a limitless enterprise for explaining everything. It certainly is (in the minds of most leading evolutionists), beginning with cosmic evolution. Even morality, which Asher assigns to religion, supposedly exists because evolution (in this case, cultural evolution) had caused its appearance.
Finally, Asher states that it does not matter if one believes in a Godlike agency behind biological diversity (p. 6). Thus, by his own standard, God is at best superfluous.
Trends in the fossil record
Asher cites a succession of first appearances, namely vertebrates, jawed vertebrates, tetrapods, amniotes, mammals, primates, and hominins, as evidence for evolution. In doing so, he completely ignores non-evolutionary explanations of these very general trends.2
In addition, by Asher’s own tacit admission, trends in the fossil record can offer only evidence for evolution that had been prefiltered through special pleading. He comments:
“At a finer scale, the story is of course more complex. Paleontologists are generally not under the illusion that we’re out to identify the literal, direct ancestor of modern groups. Nor do modern paleontologists claim that geologically older fossils always represent ancestral organisms. In fact, many fierce debates exist about the extent to which the fossil record accurately records the first appearance of a given group, and paleontologists realize that a first appearance in the rock record is an underestimate of the actual first appearance of that species on our planet” (p. 70).
Microevolution/macroevolution indistinct?
Asher argues that the boundary between micro-and macroevolution, as supposedly emphasized by creationists, is blurred (real creationists actually emphasize not small vs large but informationally uphill vs downhill). He cites the ability of bacteria to digest substances not found in nature, and the appearance of a certain protist that uses a bacterial symbiont. In doing so, he forgets that bacteria and protists follow different ‘rules’ than ‘higher’ forms of life, and so are much more versatile in terms of biologic capabilities.
The author then cites some vertebrates that have undergone remarkable changes in populations in a short period of time, namely guppies,3 stickleback fish,4 certain rodents,5 and migrating birds.6 The notion that these even approach macroevolution is laughable. Not only are all these examples of ‘tweaking’ of pre-existing traits, and furthermore that within the species, but none of them involves the addition of mutations. In fact, some of these instances can be explained by phenotypic plasticity, eliminating even a need for change in gene frequencies!
Predictions from anatomy and embryology
The author notes that Theodore Gill, back in 1872, had, using data from comparative anatomy and embryology, correctly predicted the relative ancestries of major fish groups, and done so at a time when their fossil record was virtually unknown. On the other hand, Huxley got it completely wrong on the origin of mammals. Huxley believed that mammals had originated from different groups of reptiles, and that each mammalian order, in effect, went through a premammal stage before becoming a mammal. This was parallel evolution with a vengeance!
Mammal-like reptiles, whales, and elephants
The author devotes much attention to mammal-like reptiles and whales, hailing the “incremental appearance of traits through time”. He is silent on the data manipulation and the special pleading inherent in the construction of all the cladograms.7–9
Evidently forgetting that the so-called Biogenic Law (ontogeny recapitulates phylogeny) was discredited a century ago, Asher delves into the embryonic development of the mammalian ear. He would have us believe that the progressive embryonic deployment of ear ossicles recapitulates the evolution of the post-dentary therapsid bones into mammalian ear ossicles.
One novelty of this book is elephant evolution. Being unable to read the French original, I limit my comments to his diagram (p. 114). The basal proboscideans have such short stratigraphic ranges that it is doubtful if enough specimens have been found for meaningful stratigraphic ranges. Otherwise, and contrary to evolutionary predictions, Barytherium, Primelephas, and especially Deinotherium appear later in the stratigraphic record than their more derived counterparts. Of course, as with other groups, the progression that does exist does not require an evolutionary explanation.10
Baleen whales and other toothless mammals
Several recent studies show that enamel-encoding genes in toothed animals exist as pseudogenes in toothless mammals. Asher blows the trumpet of evolutionistic triumphalism, and then pontificates that God would not make it that way (pp. 137–138). Apart from its theological presumption, Asher makes some other tacit assumptions. He supposes that, since they cannot encode for enamelin, the pseudogenes are therefore useless. Actually, there is evidence, from a more recent work,11 that at least one of the parent genes, MMP20, has multiple functions, and therefore possibly its pseudogene ortholog has at least one. If so, this ‘absolves’ God of creating non-functioning genes.
Second, the enamelin gene is just as incapable of producing enamelin if partly omitted during Creation as it is incapable of producing enamelin if omitted entirely during Creation.
Asher presumes that a de novo design is always the most intelligent solution. It may actually make more sense to create, for a toothless mammal, the same genome as for a toothed one, except for built-in intentionally inactivated enamelin genes, than to design quite different genomes for toothless and toothed mammals.
Interestingly, teeth start to develop, but later resorb, in the baleen whale embryo.12 And this should have been a clue: it has long been known that in embryogenesis one group of cells induces needed changes in other cells, a process called induction. And this is the role for these teeth. Louis Vialleton (1859–1929), who was Professor of Zoology, Anatomy and Comparative Physiology at Montpelier University, southern France, argued:
“Even though the teeth in the whale do not pierce the gums and function as teeth, they do function and actually play a role in the formation of the jaws to which they furnish a point d’apui on which the bones mold themselves.”13
Finally, the evolutionary interpretation of enamelin pseudogenes creates its own problems, unmentioned by Asher. Two different sets of the pseudogenic ‘shared mistakes’ deploy in a pattern that violates a nested hierarchy. This means that either the frameshifts occurred coincidentally and independently at the exact location twice, and that in similar fashion in two other pairs of pseudogenes, or else a ‘lineage sorting’ rationalization has to be invoked, on two separate occasions, to explain away the absence of the frameshifts at the orthologous positions of the genes in more derived genera of whales.14
DNA ‘clocks’ and DNA-based phylogenies
Asher argues that ‘clocks’ based on fossils and those based on DNA now agree. However, even if so, many molecular clocks had, in the past, contradicted those based on other evidence, and the evolutionists just explained these away by saying that molecules evolve at different rates than morphologies. No matter what turns up, evolution remains a ‘fact’.
The author also claims that degree of relatedness between mammals, as predicted by anatomical differences, agrees with that based on biomolecules. However, he notes that the inferred closeness of bats and dermopterans, 15 based on anatomy, is not supported by biomolecules (p. 176).
Consider the likelihood of similarities in substance (biomolecules) tending to correlate with similarities in construction (anatomy), regardless of origin. Why should this necessarily favour evolution as an explanation?
‘Novel information’ produced by evolution?
As if trying to impress the reader with quantity, Asher presents a long table (pp. 181–183) of items that he would have us believe are genetic and phenotypic novelties. Space limitations allow analysis of only a few of these items, none of which was chosen in a tendentious manner.
Nematodes16
A mutation is said to convert a hermaphroditic nematode into a female one. To begin with, nematodes are unusual animals with a complex sexuality, comprising hermaphrodites, pseudomales, females, etc. Second, the mode of sexuality varies according to species of Caenorhabditis and, under certain conditions, the species can interbreed and produce viable offspring. Clearly, this mutation does little more than ‘tweak’ the appearance of pre-existing sexual capabilities. No novelty here.
Simulated protein evolution17
These lab-based experiments in directed evolution (repeated rounds of mutation and selection) show how proteins can be ‘improved’ in such things as catalytic ability, binding ability, and stability. They imply that a small fraction of mutations is beneficial.
By the authors’ own admission, these experiments are of limited relevance to living systems:
“In the absence of an experimental method for measuring how changes in a protein affect biological fitness … (Also) … [experimental] mutations that benefit the target property may be selected even to the detriment of other properties … The lessons of directed evolution also caution against attributing all properties of natural proteins to adaptive causes.”18
Ironically, the simulated protein evolution shows how irreducible complexity comes into play.19 This is so, even though proteins are much simpler than such things as the human eye.
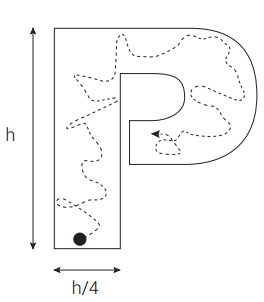
Think of natural selection acting on organisms’ phenotypes as a wind that can change speed and direction (either gradually or rapidly), or cease at any time for good (‘stasis’). The wind pushes a paintbrush on a canvas. A ‘breakthrough’ protein is produced whenever the paintbrush paints a letter. To avoid overspecifying the complexity, the letter can be any of the 26 capital letters of the English language. In addition, to avoid overspecifying the complexity, the letter does not have to be perfect (figure 1). To qualify minimally as a letter, a curve painted on the canvas must fall within a band, ¼ the height of letter thick, surrounding the actual curves of the letter in question. The outline requirement is approximately satisfied by the font Eras Bold ITC. Since natural selection cannot ‘see’ the value of the letters in advance, and partly formed letters are of no survival value to any organism, a letter will be painted only when the natural selection, for totally unrelated causes, happens to move in a way that paints a letter. Only when this occurs does the letter itself become the object of natural selection—purifying selection that will prevent overwriting of the letter, and directional selection that will eventually improve the letter to print quality.
Tooth-changing mutation20
A mutation in the Fgf3 gene causes human (and mouse) molars to be malformed, small, and lacking a hypocone.21 Far from being a novelty, this is, at best, a crude throwback to an earlier condition. To begin with, this consideration assumes evolution by invoking an Asian fossil primate, the hypocone-less Bahinina, as the ancestral state. As it turns out, this is not even a particularly good ‘evolutionary throwback’, as the resemblance of the malformed human teeth to the presumed ancestral state is only partial, and is that for which the interference of other genes is blamed.
Finally, this example is all the less impressive in view of the fact that the hypocone is believed to have appeared numerous times in different mammalian lineages, and repeatedly, even during the course of primate evolution. This is thus yet another example of so-called evolutionary convergence. These violate evolutionary nested hierarchies, and contradict the Evolutionary Tree of Life that Asher stresses so strongly throughout this book.
Gene duplication and vertebrate paired limbs22
Again, this example assumes evolution in an attempt to ‘prove’ evolution. The doubled state of the Tbx4/5 gene in vertebrates, relative to the single state of the Tbx4/5 gene in the presumed-ancestral-state exhibited by the extant cephalochordate amphioxus, is assumed to have arisen as a result of gene duplication (as opposed to separate, special creation). The ‘advanced’ morphological features that differentiate vertebrates from cephalochordates, such as migratory neural crest cells, a cranium, an endoskeleton, etc., are also tacitly assumed to have been ‘added on’ by evolutionary processes.
Experimental evidence shows that extracts from the amphioxus Tbx4/5 gene, under certain conditions, can induce limb growth in mice. This is taken as evidence that one of the factors for vertebrate limb development already existed long before their evolutionary appearance. However, what if it didn’t? Evolutionists would simply say that limb development was entirely a relatively late evolutionary development. No matter what is found, evolution is ‘true’. Interestingly, the authors betray the conjectural nature of the course of early vertebrate evolution by the very vocabulary they use (‘Proposed evolutionary scenario’, ‘following evolutionary scenarios’, ‘the first scenario’).
Vertebrate heart tissue factor23
Heart progenitor cells, in a model tunicate, are related to the actions of the lower jaw muscle and the second heart field of vertebrates. From this, a linkage is proposed in terms of evolutionary origins. However, the unmistakably storytelling character of such thinking is, again, revealed by the words used by the authors (‘has been proposed’, ‘putative heart’, ‘hints that’, ‘proposed that’, ‘could have been’).
Origin of life
Asher admits that there is currently no evolutionary explanation for the origin of life, but hastens to assert his belief that this does not make God necessary (p. 184). He then repeats the assertion that the origin of life is not part of evolutionary theory, although it is commonly called ‘chemical evolution’. What wishful thinking! If life arose from non-life, it must have undergone innumerable cycles of mutation and natural selection before it could have passed for even the simplest kind of life found on Earth today. If this was not evolution, then what was?
The ID (Intelligent Design) movement

Asher attacks the concept of intelligent design, claiming that inferences of design rest upon presuppositions of a designer. They do not. Inferences of design are empirically self-evident. For example, graffiti found on the moon would be ipso facto evidence of intelligent design, even if we have no idea about the identity or even nature of the designer, and regardless of any prior belief or unbelief in the existence of intelligent extraterrestrial life.
In stressing that evolution is non-random, in no way comparable to a tornado building something, and probable in a sense, Asher dusts off chance-parents-meeting and stadium-filling type arguments (p. 202). These naiveties completely ignore the essential difference between non-specified complexity and specified complexity.
Asher repeats the argument that exaptation of its functional components means that the flagellum is not irreducibly complex. Oh, really? The disparate parts would have to change function independently and yet in sufficiently concerted fashion so that they would be enabled to come together and begin to engage in a still-newer collective function as component members of the flagellum. How probable is that? Asher tacitly admits that the argument from irreducible complexity stands because the naysayers have not gone beyond conjecture:
“While we do not yet have a definitive, precise, step-by-step understanding of how the particular flagellum of E. coli has evolved using intermediate protein structures of some use to precursor generations (though some possibilities have been suggested) …” (p. 218).
The author attacks ID philosopher of biology Paul Nelson, insinuating that Nelson engages in dishonesty by taking advantage of lay audiences. This is offensive. I know Paul Nelson fairly well and he is a first-rate biologist. Besides, Nelson also presents his claims before very scientifically sophisticated audiences.
Academic freedom
Asher attacks evolution-questioning high-school teacher Rodney LeVake as an ‘incompetent’, and justifies the ‘discipline’ that LeVake experienced. Fortunately, Asher provides a direct quote from LeVake (on p. 223), one that makes obvious to the reader how Asher is misrepresenting LeVake. In the quote, LeVake points to the complexity of creatures in even the lower portions of the fossil record, notably right after the ‘Cambrian explosion’. Asher ignores this irrefutable fact, and instead changes the subject to various red herrings.
The author also repeats the silly argument that no religious believer in the United States has ever been sent to death, or the gulags, for his beliefs. This is patently disingenuous. For now at least, the militant enemies of Christianity, thank God, lack the political power to do any such thing! In addition, even the Communist regimes belatedly found that extreme measures are usually unnecessary (although millions of believers lost their lives because of their atheistic bigotry). Many things can have a chilling effect on free speech. For most people, the threat of a loss of reputation, status, employment, or employability is sufficient to enforce silent conformity. (In fact, one of the tenets of neo-Marxism, and even milder forms epitomized in Saul Alinsky’s Rules for Radicals, is the silencing of enemies by constantly demonizing them.) Finally, according to the Bill of Rights of the US Constitution, even a mild form of discrimination is completely unacceptable.
Conclusions
Author Robert J. Asher has written a relatively original, incisive, in-depth defense of evolution. More work is needed to evaluate his claims. However, most of his premises either fail to unambiguously support evolution or omit to mention factors that weaken, if not nullify, them.
References
- See Holding, J.P., Is the raqiya ( firmament ) a solid dome? Equivocal language in the cosmology of Genesis 1 and the Old Testament: a response to Paul H. Seely, J. Creation 13(2):44–51, 1999; creation.com/raqiya. Return to text.
- See Woodmorappe, J., Studies in Flood Geology, Institute for Creation Research, El Cajon, CA, 1999. Return to text.
- Reznick, D.N. et al., Evaluation of the rate of evolution in natural populations of guppies (Poecilia reticulata), Science 275:1934–1937, 1997. The natural selection had acted on pre-existing variation in the population. Return to text.
- Kitano, J. et al., Reverse evolution of armor plates in the threespine stickleback, Current Biology 18:769–774, 2008. This was a reacquisition of a once-common trait in the not-distant past, caused by nothing more than the change of the frequency of a preexisting allele in the population. Return to text.
- Pergams, O.R.W. and Lawler, J.L., Recent and widespread rapid morphological change in rodents, PLoS ONE 4(7):e6452, 2009. The authors note that these changes can be explained by phenotypic plasticity rather than changes in gene frequency (which itself could have originated from a rare pre-existing genotype instead of a mutation), and admit that, “Alternatively, we have noted phenotypic plasticity as another potential mechanism of rapid morphological change, and plasticity is notoriously difficult to distinguish from direct genetic evolution, especially in historical samples” (p. e6452). Return to text.
- Pulido, F. and Berthold, P., Current selection for lower migratory activity will drive the evolution of residency in a migratory bird population, PNAS (USA) 107(16): 7341–7346, 2010. The authors detect a change in migration caused by genetics (selective breeding). They note that few studies have shown that changes in organisms in relation to climatic change have demonstrated a genetic basis, as opposed to phenotypic plasticity. Return to text.
- Woodmorappe, J., Walking whales, nested hierarchies, and chimeras: do they exist? J. Creation 16(1):111–119, 2002; creation.com/walkingwhales. Return to text.
- Woodmorappe, J., Mammal-like reptiles: major trait reversals and discontinuities, J. Creation 15(1):44–52, 2001; creation.com/mammal-like. Return to text.
- Woodmorappe, J., Bird evolution: discontinuities and reversals, J. Creation 17(1):88–94, 2003; creation.com/bird-evolution. Return to text.
- Actually, any progression is very crude once the uncertainties of stratigraphic ranges are considered. I looked up all proboscidean genera in the Paleobiology Database (paleodb.org) and printed out their stratigraphic ranges, complete with the stratigraphic ranges expanded to their respective 95% confidence limits. This is according to the contained built-in statistical methodologies and assumptions of Strauss and Sadler, Marshall, and Solow. According to the results of all three methods, there is a very great deal of statistical overlap of the first appearances of ‘primitive’ through ‘derived’ genera. For instance, the first appearance of Elephas, to which the modern Indian elephant is referred, actually overlaps all (or all except one) of the known proboscidean genera, including those featured by Asher (p. 114)! How many other well-known fossil ‘series’ suffer from the same fundamental problem? Return to text.
- Meredith, R.W. et al., Pseudogenization of the tooth gene enamelysin (MMP20) in the common ancestor of extant baleen whales, Proc. Royal Society B 278:993–1002, 2011. Return to text.
- Demere, T. et al., Morphological and molecular evidence for a stepwise evolutionary transition from teeth to baleen in mysticete whales, Systematic Biology 57(1)15–37, 2008. Return to text.
- Vialleton, L., L’origine des Êtres Vivants [The origin of living beings], Librarie Plon, Paris, 1930. Return to text.
- Meredith, R.W. et al., Molecular decay of the tooth gene enamelin (ENAM) mirrors the loss of enamel in the fossil record of placental mammals, PLoS Genetics 5(9):e1000634, 2009. See especially figure 1. Return to text.
- Meaning ‘skin-winged’, they are the colugos, also called ‘flying lemurs’, although they are really gliders. See also Catchpoole, D., The Colugo Challenge: Perfectly fit for its life in the trees, the colugo just doesn’t fit into the evolutionary tree of life , Creation 33(2):28–31, 2011; creation.com/colugo. Return to text.
- Baldi, C. et al., Mutations in two independent pathways are sufficient to create hermaphroditic nematodes, Science 326:1002–1004, 2009. Return to text.
- Bloom, J.D. and Arnold, F.H., In the light of directed evolution: Pathways of adaptive protein evolution, PNAS (USA) 106(Supp. 1):9995–10000, 2009. Return to text.
- Bloom and Arnold, ref. 17, p. 9999. Return to text.
- In order for natural selection to make the ‘first step’ in selecting in favour of a protein capability, a fortuitously pre-existing ‘promiscuous activity’ of the protein must be present. Also, directed evolution is stymied by local fitness peaks, where no further incremental improvement is possible. This can be overcome by ‘not setting the bar too high’ and, specifically, by ‘identifying appropriate intermediate challenges’ (Bloom and Arnold, ref. 17, pp. 9997–9998). By whom? By the experimenter! Otherwise, since evolution lacks foresight, this comparatively mild challenge of irreducible complexity can only be overcome by a set of natural-selection events that, while not fortuitous in themselves, act in fortuitously proper sequence and timing to change natural selection in just the right way to eventuate in a ‘breakthrough’ protein. Return to text.
- Charles, C. et al., Modulation of FGf3 dosage in mouse and men mirrors evolution of mammalian dentition, PNAS (USA) 106(52):22364–22368, 2009. Return to text.
- One of the cusps on the molar tooth in hominids. Return to text.
- Minguillon, C. et al., Tbx4/5 gene duplication and the origin of vertebrate paired appendages, PNAS (USA) 106(51):21726–21730, 2009. Return to text.
- Stolfi, A. et al., Early chordate origins of the vertebrate second heart field, Science 329:565–568, 2010. Return to text.
- Since coming up with the foregoing exercise, I have learned that others have developed more sophisticated exercises that illustrate the nature of irreducible complexity. The irreducible complexity of Chinese Han characters is comparable to that of folds in protein: Axe, D.D. et al., Stylus: A system for evolutionary explanation based on a protein proteome model with non-arbitrary functional constraints, PLoS ONE 3(6):e2246, 2008. See also Axe, D.D. et al., A Stylus-generated artificial genome analogy to bacterial genomes, Bio-complexity.org 3(2)1–15, 2011. Return to text.





Readers’ comments
Comments are automatically closed 14 days after publication.