Journal of Creation 25(2):22–31, August 2011
Browse our latest digital issue Subscribe
Did birds evolve from dinosaurs?
A review of the popular dinosaur–bird link shows that the case for feathered dinosaurs is mixed, with some claimed ‘protofeathers’ possibly being fossilized features of the skin. Some claimed theropods did have true feathers, some even with flight feathers on their feet, but there are questions as to whether these creatures are really dinosaurs or are unique, extinct birds similar to Archaeopteryx. Furthermore, careful reassessment shows that the popular belief in the dino-to-bird transition is based on a flawed cladistics analysis.

The idea that birds evolved from dinosaurs has excited the evolutionary imagination for many years. Thomas Huxley, called Darwin’s Bulldog because of his aggressive promotion of evolution, was the first to suggest an evolutionary connection between dinosaurs and birds, an idea that remained popular in the 1800s.1 However, this idea waned in the early 1900s because paleontologists believed dinosaurs were too specialized for birds to have arisen from that line. By ‘specialized’ they meant that dinosaurs had evolved too many specific complex features for birds to have evolved from them. The avian origin from reptiles was still accepted, but the parent creature was thought by some to be a crocodile-like animal, and by others to be from the flying reptiles, the pterosaurs.
In about 1980, the dinosaur–bird connection once again became popular. It started with the idea that dinosaurs may have been warm blooded.2 The theory has been fuelled by numerous discoveries of dinosaurs, birds, mammals, and other creatures in Liaoning Province, northeast China. Among these fossils were at least nine that paleontologists claimed were feathered theropod dinosaurs. This has been widely reported and promoted by the media and popular science journals. Even the journal Nature cited these discoveries as proof that birds evolved from dinosaurs.3 It is even suggested that some tyrannosaurids (similar to, but not quite like, T. rex) had protofeathers4,5 Museum displays are now adding feathers to dinosaurs where there is no evidence for feathers, such as the display in the Museum of the Rockies, Bozeman, Montana, showing a feathered Deinonychus attacking a duck-billed dinosaur (figure 1).
However, these claims have met with controversy within evolutionary circles. Creationists have analyzed these arguments in detail and found them problematic.6–9 This issue of feathered dinosaurs is confusing, partly because we do not have all the information, and future discoveries will undoubtedly change the picture. At present, however, enough is known to question the reality of ‘feathered’ dinosaurs and the avian descent from these reptiles.
The origin of feathers from scales or some other part of the skin is an extremely difficult evolutionary problem.10 Feathers seem simple but are very complex.7,11 Furthermore, the whole anatomy and physiology of the evolving creature must be radically changed. The evolution of powered flight also presents a major obstacle. Even those who have implicit faith in the reality of evolution have noted these difficulties:
“In either case, the anatomical changes needed for flying must have evolved in a sequence of very small steps, because nothing we know about evolution allows us to believe that feathered wings could have appeared abruptly as an innovation in avian anatomy. Wings must have evolved over a very long time span. And each new modification of body plan or limbs during that period must have made some contribution to fitness long before the day when a jumping or gliding creature gave the first strong beat of its forelimbs and ceased simply falling back to earth.”12
Prior to 1980, it was argued that birds had not evolved from dinosaurs because dinosaurs lack a furcula, or a wishbone.13 Since then, it has been discovered that some dinosaurs do have furculas of various types.14–19 This discovery helped tilt opinion towards the dino-bird link.20 Another discovery supporting the avian descent from dinosaurs was that some dinosaurs had hollow bones, generally considered a bird trait.21,22 But the most convincing argument for evolutionists has come from the new classification system known as cladistics. Therefore, I will summarize each of these recent discoveries and ideas, demonstrating that the evidence is not as strong as many think.
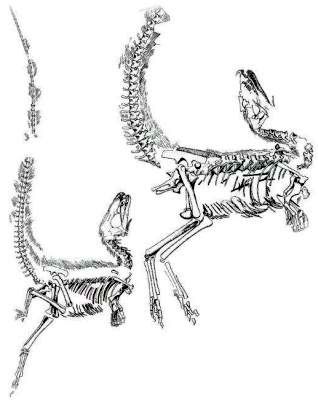
Claimed feathered dinosaurs from China
All the discoveries of ‘feathered’ dinosaurs have been made in northeast China.22–25 There seems to be two groups of feathered dinosaurs: those with ‘protofeathers’ and those with true feathers. Sinosauropteryx prima is claimed to have ‘protofeathers’, dubbed ‘dino fuzz’ by some, as shown by fibres that emanate from the skeletons (figure 2).22,23 Such fibres are also found on Sinornithosaurus millenii from China.26 They have been called integumentary (skin) structures. Recently, colour pigments have been found in the integumentary structures of Sinosauropteryx that lend support to the belief that these structures are decayed feathers.27,28
Caudipteryx, Protarchaeopteryx, Microraptor and a few others are claimed to have true feathers of modern aspect and are classified as small theropod dinosaurs. Microraptor actually has four wings, with the second pair coming off the hind legs.29
Such data has been hailed as infallible proof that dinosaurs evolved into birds.22 Paul Sereno stated in 1997 that the origin of birds from dinosaurs has been resolved in its favour.30 Phil Currie has no doubts and believes that theropod dinosaurs are alive today:
“In fact, because birds are the direct descendants of theropods, they are technically classified as theropod dinosaurs. Therefore, there are more than 10,000 species of theropods alive today.”31
Currie believes that many dinosaurs had feathers.32 Others are even more dogmatic: Richard Prum is adamant that birds are modern dinosaurs, claiming that those who disagree with him are non-scientists.33 In doing so, he dismisses all the critical evidence against his position. Prum believes the new information has ‘redefined’ the science of ornithology: “The recognition that birds are theropod dinosaurs has redefined the science of ornithology as extant dinosaur biology.”34
As a result of this new theory, scientists have ‘discovered’ many behaviours in birds that they believe can be traced back to their dinosaur ancestors, such as parental care and brooding. These links are supported by findings of adult dinosaurs fossilized in close association with egg clutches.35 However, brooding is not unique to birds, and claims that this fossil evidence demonstrates brooding go beyond the evidence, since they were not in a brooding pose above the eggs.36,37 There are other possibilities; e.g. the fossil dinosaurs could have died while laying their eggs.
Problems with ‘feathered dinosaurs’
In spite of recent discoveries and the enthusiasm for the dinosaur ancestry of birds, significant evidence suggests that the question remains open to doubt among many evolutionists. Ornithologist Alan Feduccia and others have challenged all the claims of feathered dinosaurs. However, part of their opposition is based on their bias that birds evolved from different reptiles that lived before the dinosaurs, although Feduccia admits that there is little or no evidence for this hypothesis, either.38 Another possibility, of course, is that neither hypothesis is valid because evolution has not happened. We need to examine some of the other problems with the new discoveries.
Another reason for skepticism should be the discovery that some of the fossils promoted as proof turned out to be fraudulent, cobbled together to look like a missing link between dinosaurs and birds. Archaeoraptor—which made the front cover of National Geographic in November 1999—was hailed as ‘proof’ of feathered dinosaurs and the dinosaur–bird link. However, like so many transitional fossil ‘proofs’, it was subsequently shown to be a forgery.39 It turned out to be a fossil bird with a dinosaur tail glued on. Most telling was that this simple deception succeeded in fooling many scientists.
Bird evolution from a lizard-hipped dinosaur?
Dinosaurs have long been classified by the structure of their hip; the two branches are the lizard-hipped dinosaurs and the bird-hipped dinosaurs.40 Interestingly, it appears that bird evolution was from the wrong type—the lizard-hipped theropods. Barrett noted: “Confusingly, bird-hipped dinosaurs are only distantly related to birds, whereas the direct ancestors of birds are to be found among the lizard-hipped dinosaurs.”41
DeCourten corroborated this observation:
“Confusingly, though, these bird ancestors were members of the order Saurischia [lizard-hipped dinosaurs], not the Ornithischia [bird-hipped dinosaurs], as we might expect from the names alone.”42
So, evolutionists are forced to believe that hips diagnostic of birds evolved at least twice, despite the complete lack of fossil evidence for such an evolutionary swerve.
Integumentary structures likely not protofeathers
Although it is possible that the integumentary structures are decayed true feathers, there are some problems with that deduction. Feduccia and others have analyzed the claims of the feathered dinosaurs from China in some detail.43 They found experimental and fossil evidence that the fibres found along the outside of the skeleton of Sinosauropteryx and others are better explained as collagen fibres that were simply a part of the skin of the dinosaur.44–46
We also know that there are several dinosaurs with no claimed evolutionary link to birds that also have these fibres. One of these is Psittacosaurus, a horned dinosaur similar to ceratopsians.43,45,47,48 A second is a new birdhipped dinosaur found in China that incidentally extended the stratigraphic range of this particular kind of dinosaur up towards the present by at least 60 million years,49,50 illustrating, if nothing else, that theoretical changes may be demanded by new evidence waiting to be discovered. Birds did not supposedly evolve from bird-hipped dinosaurs, but from lizard-hipped dinosaurs (see above), which is why the discovery of integumentary structures on a bird-hipped dinosaur is so significant.
Other types of fossil animals also show these bristlelike collagen fibres, such as a pterosaur, an ichthyosaur, and other reptiles and dinosaurs.51 Even collagen fibres from a dead dolphin were similar to the claimed protofeathers from China.52 Thus, these fibrous structures cannot be considered unique to dinosaurs in the evolutionary chain leading to birds, which also supports the conclusion of Feduccia et al. that they are simply collagen fibres.43 For those who believe in dinosaur-bird evolution, the picture has become fuzzier:
“Perhaps the only clear conclusion that can be drawn from the foregoing is that little Tianyulong has made an already confusing picture of feather origins even fuzzier. Such an outcome is common in palaeontology.”53
It may be that the colour pigments found in the integumentary structures of Sinosauropteryx are not unique to feathers, but this is disputed by Lingham-Soliar.48,54 Lingham-Soliar summarizes by concluding, after much research, that the evidence for protofeathers is non-existent:
“It is important to understand that this paper neither supports nor refutes the hypothesis of protofeathers but rather rejects the alleged evidence from fossils to date.”55
The temporal paradox
Another more minor problem with the ‘feathered dinosaurs’ from China is the reported age of the new fossils. They are mostly 25 Ma younger than the first true bird fossil, Archaeopteryx, which was found in the Late Jurassic, which is supposedly 150 Ma ago.56 Scientists tend to come up with complex terms for simple issues, and this is no exception; geologists call it the ‘temporal paradox’. However, supporters of the dinosaur-bird link have a point when they claim that the paradox is not significant because the ‘feathered dinosaurs’ from China and the first true bird could have diverged from a common ancestor over 150 Ma ago.22,57 But Feduccia noted that Archaeopteryx has such well-developed feathers and other avian anatomical features that the evolutionary origin of birds must be much earlier than 150 Ma.51
And as expected, the temporal paradox is believed to have been solved just recently, but not without a cost. A crow-sized feathered ‘theropod’, Anchiornishuxleyi, was found in strata dated 151 to 161 Ma old—before the age of Archaeopteryx at 150 Ma old.58,59 The problem with this new feathered dinosaur is that it had feathers on its legs, like Microraptor.
Another bird-like creature with feathers on its feet, which is a little older than Archaeopteryx and is called Pedopenna, has also been found.60 Although some birds have feathers on their legs today, such as some pigeon breeds, they are not flight feathers as those on Anchiornis, Microraptor, and Pedopenna. These three creatures are now classified into a basal group of theropods called Paraves.
Microraptor has always been dated significantly younger than Archaeopteryx. However, the early dates, before Archaeopteryx, on Anchiornis and Pedopenna are actually poorly resolved.61 But a new find of an alverezsauroid, dated at 63 Ma older than previous fossils, supposedly rescues the ‘solution’ to the temporal paradox. In Science, the creature is depicted with feathers.27 However, the accompanying article says nothing about feathers, so this creature cannot be called a feathered dinosaur. It seems the temporal paradox may have been ‘solved’ by dating manipulations.
So, in ‘solving’ the temporal paradox, evolutionists have opened up more conundrums within their paradigm. The new discovery shows that feathers and other bird-like features could be older than Archaeopteryx. Secondly, it appears that the evolutionists have a more convoluted origin of flight.59 Feathers, they say, first evolved on all four limbs and then disappeared on the hind limbs over time. Then some of these feathered dinosaurs even lost the ability to fly and became secondarily flightless birds. Anchiornis also had slender limbs causing the original researchers to conclude that this animal was adapted to running.62 How would these creatures run with flight feathers on their feet? And if the evolutionists go further back in their time scale, evolution becomes very complicated. The earliest dinosaurs are far from bird-like; they are too specialized.63 Then some of these specialized first dinosaurs had to evolve feathers and other bird-like features, only to lose them again as small theropods evolved from these feathered dinosaurs.
Same type of dinosaur, but no feathers
Many of the dinosaurs thought to be feathered have been found outside China lacking the expected feathers. For example, a new dinosaur found in the Solnhofen limestone in Bavaria—the formation that produced the best Archaeopteryx fossils—comes from the same group that was reported to possess ‘protofeathers’ in China.64 There were no integumentary structures and: “The discovery will encourage a re-evaluation of feather evolution.”65 Not only that, but many of the ‘feathered dinos’ coming out of the Jehol group in China have no feathers (or ‘integumentary structures’) either: e.g. Mahakala,66 Gigantoraptor,6 and Velociraptor.67 Feathers are often assumed a priori without evidence. Since ‘feathered dinos’ are so controversial they should have clear evidence of feathers before being admitted into the discussion. Cladistic similarity is not enough.
Dinosaur lungs more like those of reptiles
It takes more than feathers to make a bird. Dinosaurs would have had to evolve a variety of unique structures along the path to true birds. A major hurdle is the development of an avian respiratory system. Dinosaur lungs, including those of the theropods, which some paleontologists believe evolved into birds, were unique. Based on hollow air spaces in some theropod dinosaur bones, some evolutionists have tried to claim that these dinosaurs had lungs like birds.68,69 Such air spaces are equivocal as far as whether these dinosaurs had bird-like lungs.70 However, others believe the theropod lungs resembled those of modern-day crocodiles, not birds.71,72 This also implies that theropod dinosaurs were ectothermic (cold-blooded), providing another hurdle on the evolutionary trail to birds. It is difficult to even imagine how an ectothermic animal with lungs like a crocodile could have evolved into a bird with their clearly divergent respiratory and metabolic systems:
“The evolutionary implications are even more far-reaching. Ruben argues that a transition from a crocodilian to a bird lung would be impossible, because the transitional animal would have a life-threatening hernia or hole in its diaphragm.”73
After twelve years of further research, Quick and Ruben recently conclude that the theropod breathing apparatus was like a crocodile and unlike birds:
“We conclude that there are few data supportive of there having been an avian style lung airsac system in theropods or that these dinosaurs necessarily possessed cardiovascular structure significantly different from that of crocodilians. These conclusions are reinforced by previously cited evidence for crocodilian-like lung ventilation in theropod dinosaurs (Ruben et al., 2003).”74
Advocates of the dinosaur-to-bird evolutionary path have argued that the barriers are not impossible to climb, but their argument appears quite convoluted.75
Dinosaurs not only possessed lungs like crocodiles, but possessed the narrow noses of these reptiles, which is consistent with low lung ventilation.76 Nasal passages of dinosaurs are small, only a quarter as large as those of warm-blooded animals. This supports the conclusion that dinosaur metabolism was significantly lower than that of birds or mammals.
Bird and dinosaur limb digits differ in their development
Evolutionists argue for a link between dinosaurs and birds based on comparative morphology, even during embryonic development. Birds and dinosaurs have similar features, called ‘shared derived characteristics’ (see cladistics analysis below). However, a major morphological feature supports an opposite conclusion. During embryonic growth, bird limb digits developed from a five-finger arrangement into one that has only the second, third and fourth digits (II, III, IV). On the other hand, dinosaur growth exhibits a different embryonic pattern—I, II, III.77–79 The bird trend in the past had been a little uncertain but has been verified.80,81 This is powerful evidence as adduced by paleontologists.
One obvious interpretation of these data is that birds did not evolve from dinosaurs. However, those who refuse to accept that conclusion have developed the ‘frame-shift hypothesis’—part of avian evolution included a frame shift in the expression of the digits.82 During the transitions from dinosaurs to birds, the embryonic expression somehow shifted from I, II, and III to II, III, and IV. Unfortunately, bias and circular reasoning permeate this hypothesis:
“One solution to the problem of avian digit homology [similarity] in favor of the II, III, IV hypothesis is to assume that birds are not theropod dinosaurs … Removing birds from theropods implies that the similarities between the hands of Archaeopteryx and that of theropods are convergent and not homologous. This is, however, not a very satisfactory explanation for the available data [emphasis mine].”83
‘Frame shift’ advocates point to a theropod fossil discovered in China that hints at such a shift.84 But the dinosaur in question not only reduced digit I, but also IV and V, so that it only had two long digits. Proponents cannot find an ‘adaptive’ reason for the proposed shift either.81 I.e. there is no evolutionary advantage that would accrue from this shift. Nor is there any advantage to the intermediate steps required by this shift.
Feduccia asserts that there is no evidence for the frameshift hypothesis.51 James and Pourtless IV conclude that it was simply made up to explain away the digit contradiction by those that were adamant that birds evolved from dinosaurs:
“ … these data do not alter the logical status of the frame-shift hypothesis as an ad-hoc auxiliary hypothesis … It was introduced for the explicit purpose of restoring agreement between predictions of the BMT [birds are maniraptoran theropod dinosaurs] hypothesis and repeatedly obtained falsifying observations.”85
Despite all this, Chiappe steadfastly maintains his faith in a dinosaur origin for birds, but his analysis is not convincing.86 This type of reasoning is why many critics say evolution cannot be falsified.
‘Dinosaurs’ with true feathers are birds
Some researchers now embrace the idea that the small ‘theropod dinosaurs’ with true feathers, especially Microraptor, are really birds,43,87,88 probably flightless.43,89 In fact, when first discovered, some were classified as birds. Scientists first thought Caudipteryx was a bird.90 Scientists once believed that alvarezsauroids were flightless birds but now believe they are theropods.27 Wellnhofer states that the classification is “basically a problem of definition that possibly may never be resolved.”91 If this is true, then the avian-dinosaur link and even the existence of ‘feathered’ dinosaurs are called into question.
In a more sophisticated cladistic analysis (see below), James and Pourtless IV reach the same conclusion: that the ‘feathered’ dinosaurs are in fact birds.92 Feduccia et al. related how one Chinese dinosaur-to-bird advocate admitted that Caudipteryx and Protarchaeopteryx could be birds:
“Xu et al. (2001, p. 200) concede that two taxa with true feathers, Caudipteryx and Protarchaeopteryx, are controversial insofar as they have been proposed to be flightless birds rather than dinosaurs and that confirmation of the theropod origin of feathers requires documentation of unambiguously feather-like structures in a clearly non-avian theropod.”93
It is more than possible that the early excitement of finding ‘feathered’ dinosaurs caused the case to be overstated, and that the strange fossils found in China are quite likely to be extinct, unique birds, similar to Archaeopteryx in a number of features.
There are also bird fossils found with the ‘feathered dinosaurs’,94 making it more likely that the creatures that possessed true feathers were indeed birds. Microraptor also had bird teeth and not dinosaur teeth, reinforcing this view.95 And the new Anchiornis also had bird-like teeth in their being unserrated.59 Xu et al. also originally classed Anchiornis as a bird.96
Archaeopteryx is a unique bird—so no missing link
Archaeopteryx was first reported in 1861 within fine-grained limestone in Bavaria (figure 3). The name means ‘ancient wing’ and the creature is said to be 150 Ma old.97 Archaeopteryx is considered the perfect missing link:
“The Archaeopteryx fossil is, in fact, the most superb example of a specimen perfectly intermediate between two higher groups of living organisms—what has come to be called a ‘missing link’, a Rosetta stone of evolution.”98

Since then, a total of ten specimens have been found, if the isolated feather (which is considered too small to be from Archaeopteryx by Wellnhofer99) is counted as one. The tenth fossil was reported in 2005.100,101
There is no doubt that Archaeopteryx is unique. It had a generally reptilian skeleton with a tail made up of extended vertebrae, teeth, and claws on its wings. But it also had 100% modern feathers showing the asymmetry of flight feathers. It is considered the first bird, but those advocating the dinosaur–bird link want to make Archaeopteryx a feathered dinosaur. In fact many of the so-called feathered dinosaurs resemble Archaeopteryx.
Many evolutionists emphasize its reptilian features, but these are overstated.102 For instance, no modern birds have teeth, but some fossil birds do. Even some dinosaurs did not have teeth, such as Oviraptor and the man-sized ornithomimids.103,104 Also, its teeth are bird-like and not those of reptiles.95 Thus, the teeth are not conclusive, and being an extinct bird, this is not surprising. Feduccia stated that “Archaeopteryx was much more birdlike in many features than has been previously thought.”105
Its wing claws are not indicative of reptilian status either. Some young birds, especially flightless birds, have claws on their wings when young, including the hoatzin and the young of the touraco.87,106
Its tail was reptilian, but a number of extinct birds, including some of the ‘feathered dinosaurs’, have long tails that are extensions of their vertebrae, like Archaeopteryx.
Claws like modern perching birds
Other features place this fossil squarely within the family of birds. Archaeopteryx had bird-like claws. Feduccia analyzed the curvature of the hind claws and concluded that their curvature was similar to claws in modern perching birds.107–109 Birds can have less curvature, but these are invariably ground dwellers. Archaeopteryx would have been ill-suited for running, but well suited to gripping tree limbs. Its wing claws may have been used to climb tree trunks, like a woodpecker.
Several dinosaur–bird enthusiasts dispute this analysis, saying that the Archaeopteryx hallux (the first toe which is opposed to the other three) is less than an optimal 180° in the tenth specimen, making it a poor percher.100,101 However, modern perching birds exhibit hallux angles from 65°–180°.56 Also, Mayr et al. ignored the curvature of the claws.100,101
Brain and inner ear like a bird
An analysis of the brain and inner ear of Archaeopteryx showed that they were similar to birds:
“Here we show the reconstruction of the braincase from which we derived endocasts of the brain and inner ear. These suggest that Archaeopteryx closely resembled modern birds in the dominance of the sense of vision and in the possession of expanded auditory and spatial sensory perception in the ear.”110
It could fly well
Advocates of avian evolution from dinosaurs claim that Archaeopteryx could not fly well because it lacked powerful flight muscles and that its primary four feathers were not asymmetrical.111,112 Some even believe that Archaeopteryx was adapted to running and taking off from the ground. But its wing and tail feathers were strongly asymmetric—well within the range of modern birds that flap their wings.113–115 Their argument is also contradictory because more powerful flight muscles are needed to take off from the ground than from trees: “Both anatomy and phylogeny strongly suggest that Archaeopteryx was an arboreal bird.”116 Given its many ‘adaptations’ for flight, one would expect that it flew well.117 Archaeopteryx not only looks like a modern bird in many ways, but evidence “suggests that Archaeopteryx had an advanced aerodynamic morphology.”118 Its feathers are indistinguishable from those of modern birds:
“Regardless of the degree to which Archaeopteryx’s skeleton was reptilian, there can be no doubt that its feathers were indistinguishable in any important ways from those of living birds. Its wings had the basic pattern and proportions of the modern bird’s wing; indeed, there has been no essential change in this aerodynamic structure for about 150 million years.”119
Even Chiappe admits: “… its feathers are already differentiated into structurally modern types.”120
Why believe in the dino–bird link?
Given this evidence, why then do many paleontologists remain dogmatic about dinosaurs evolving into birds? The main reason, even before the discovery of ‘feathered dinosaurs’, is their analysis of comparative anatomy using a relatively new classification scheme called cladistics that puts birds together with theropod dinosaurs.121 Witmer wrote:
“Birds are dinosaurs. That’s hardly the stuff of headlines any more, as data have streamed in revealing anatomical similarities [from cladistics] between birds and the theropod dinosaurs from the tips of their noses to the tips of their feathered tails.”122

Cladistics is a method that classifies organisms in a nested hierarchy of similarity based on a comparison of individual characteristics. It also depends on objective, quantitative analysis, employing computers. When comparing fossils, the program uses a binary system of either yes (1) or no (0) for a particular feature or characteristic that either exists or does not exist. Many traits are compared in the analysis. The more that score (1), the more closely related the subjects are assumed to be. Scientists then build cladograms (figure 4) and infer evolutionary relationships from them. Practitioners of cladistics dismiss their critics, such as Alan Feduccia, Larry Martin, and Storrs Olsen.123 They claim that the many shared-derived characteristics in birds and theropod dinosaurs could not happen by chance.81 This is ironic, given that chance is fundamental to evolution (random mutations are its engine), and, of course, common design explains the shared traits quite readily.
Evolutionary cladistics presents itself as objective science, yet is very subjective. A major problem is that one must ‘know’ ahead of time which traits have real evolutionary significance, rather than having been caused by ‘convergent evolution’. Traits believed to be the result of evolution are called ‘shared-derived characters’. Convergent evolution is the evolution of similar structures in similar environments by organisms that are not directly related by evolution, such as flight in birds, bats, reptiles, and insects. It is assumed that similar environments resulted in all the anatomical modifications required for flight. Ironically, evolutionists admit that there are more traits developed by convergence than by true evolutionary descent. In fact, ‘convergent’ evolution seems little more than an ad hoc explanation for many evolutionary puzzles:
“It is difficult for me to theoretically understand how convergent evolution could work, due to all the many subtle differences between present similar environments, the rarity and randomness of mutations, the lack of direction and the conservative nature of natural selection, and the multitudinous pathways that organisms could have taken.”124
Ronald Jenner has subdivided cladistic analysis into three steps: (1) the analysis of the morphological data, (2) the computer program, and (3) the results in the form of a cladogram that supposedly shows evolutionary relationships (figure 4).125 Jenner states that cladistics, as currently practiced, is seriously flawed:
“Uncritical and nonexplicit character selection, character coding, and character scoring seriously compromise Step 1 … . Failure to identify problems and errors generated in Step 1 of the research cycle is testament to the general failure of Step 3.”126
However, the problem goes deeper. The cladograms and mathematical analyses involved in cladistic analysis gives paleontology an air of objectivity it has no right to claim. Paleontology always works with vastly incomplete morphological data, which means Jenner’s step 1, the analysis of morphological data, will always be uncertain and subjective. Burke and Feduccia stated: “The conflict pivots on the significance awarded to different types of data in the identification of homology.”127 Homology is any similarity between characteristics of organisms that is due to their shared ancestry through evolution. Feduccia added:
“Another way of stating the problem is that cladistic methodology in paleontology has forced into algorithmic form what is arguably the most subjective and qualitative field in biology.”128
Because of the incomplete nature of paleontological data, there is never any certainty of whether traits are due to descent or convergence. It often becomes nothing more than an expression of the bias of evolutionists.75 This means evolutionary cladistics is essentially circular reasoning. Ornithologist Larry Martin was blunt in his assessment:
“As for the many cladograms that demonstrate a dinosaurian origin of birds, Martin charges that they are riddled with characters based on mistaken anatomy—in other words, ‘garbage in, garbage out’ on a massive scale.”129
Jenner infers that the major problem is that not all anatomical information goes into the computer analysis.125 He strongly recommends that all anatomical information should go into cladistics analysis and that a conscious effort to minimize bias should be developed.
James and Pourtless IV did just this in a cladistic analysis of the dino–bird link, concluding that a dinosaur to bird path was not strong.92 Birds could just as easily have evolved from crocodiles or other extinct reptiles.
Of course, creationists have no trouble explaining similarities, using the concept of common features resulting from common design, thus avoiding the messy uncertainty of ‘real’ vs convergent evolution. The regularity of ‘convergence’ in evolutionary explanations testifies to this: there is similarity in many biological traits, with enough divergence in specifics to thwart common origin as an explanation. Just as man-made artifacts have similarities due to design parameters, so too do creatures. Different models of cars share many ‘traits’ because they are intended to do the same thing. Each one possesses tires, an engine, steering, windshields, lights, etc. Differences show variations in purpose: an off-road truck is not the same as a family sedan. This is similar to what we see in the biological world, which can be ascribed to a great Creator.
Summary
Secular paleontologists argue whether or not birds could have evolved from dinosaurs. But these paleontologists all have one thing in common; they refuse to debate their assumption that evolution occurred. Consistent with the view that it has not, we agree with the arguments that there is no compelling evidence for the dinosaur-to-bird evolutionary path:
“However, answers to the question of the immediate ancestor of birds remain elusive, as does the overall early radiation from the Dinosauromorpha.”130
Birds did not evolve from dinosaurs; they did not evolve from anything. Feduccia et al. admit: “The major problems related to the origin of birds are still unresolved.”95 Feduccia further admits that there are no other ancestor candidates for the evolution of birds:
“Paleontological cladists claim that opponents of the theropod origin must produce a more suitable ancestor, but alas, we simply don’t have sufficient evidence. We can only say, as dictated by science and logic, that the ancestor was surely a small, quadrupedal, arboreal archosaur [extinct reptile], a pre-dinosaur in the overall scheme of the genealogy.”38
Thus, even the experts acknowledge that the origin of birds is still unknown. This is surprising, given the amount of time, man-hours, and money that has been poured into research over many decades. Feduccia is puzzled:
“What’s the problem? We have some of the best-preserved fossils in the entire vertebrate series in the seven skeletons of Archaeopteryx, we have a wonderfully preserved array of fossil reptiles from the Triassic, Jurassic, and Cretaceous periods, and we have scores of well-educated scientists working on the problems of avian relations. So why isn’t the problem resolved after much more than a century?”131
There is an easy answer to that question, though one not accepted by secular scientists because it questions their presuppositions, which are generally held by faith rather than evidence. The answer is to reject the constraints of the worldview of naturalism and to jettison faith in evolution, to look at the actual evidence that shows discontinuity between major groups, and to admit the likelihood of creation.
Feduccia and other ornithologists who say that there is no evidence for the dinosaur–bird link are correct. Their opponents, who argue that there is no evidence for the evolution of birds from extinct reptiles, are also correct. Both are correct because evolution down any path never happened. Observation and reason both support an original creation of different kinds of organisms (Genesis 1).
Acknowledgements
I thank John Reed for reviewing an earlier manuscript and offering valuable comments. I also thank Ray Strom for his two digital pictures from the Museum of the Rockies.
References
- Chiappe, L.M., Glorified Dinosaurs: The Origin and Early Evolution of Birds, University of New South Wales Press, Sydney, Australia, 2007. Return to text.
- Feduccia, A., The Origin and Evolution of Birds, second edition, Yale University Press, New Haven, CT, p. 66, 1999. Return to text.
- Chiappe, ref. 1, p. 71. Return to text.
- Chiappe, ref. 1, p. 64. Return to text.
- Xu, X., Norell, M.A., Kuang, X., Wang, X., Zhao, Q. and Jia, C., Basal tyrannosauroids from China and evidence for protofeathers in tyrannosauroids, Nature 431:680–684, 2004. Return to text.
- Doyle, S., Big birdosaur blues: new fossil creates problems for dino-to-bird evolution, Journal of Creation 21(3):5–6, 2007. Return to text.
- Menton, D., Formed to Fly, AiG DVD, Hebron, KY, 2007. Return to text.
- Sarfati, J., Ostriches break dino-to-bird theory, Creation 25(1):34–35, 2003. Return to text.
- Woodmorappe, J., Bird evolution: discontinuities and reversals, Journal of Creation 17(1):88–94, 2003. Return to text.
- Bergman, J., The evolution of feathers: a major problem for Darwinism, Journal of Creation 17(1):33–41, 2003. Return to text.
- Lingham-Soliar, T., Bonser, R.H.C. and Wesley-Smith, J., Selective biodegradation of keratin matrix in feather rachis reveals classic bioengineering, Proceedings of the Royal Society B 277:1161–1168, 2010. Return to text.
- Feduccia, ref. 2, p. 93. Return to text.
- Hinchliffe, R., The forward march of the bird-dinosaurs halted? Science 278:596–597, 1997. Return to text.
- Barrett, P., National Geographic Dinosaurs, National Geographic Society, Washington, D.C., p. 176, 2001. Return to text.
- Burnham, D.A., New information on Bambiraptorfeinbergi (Theropoda: Dromaeosauridae) from the late Cretaceous of Montana; in: Currie, P.J., Koppelhus, E.B., Shugar, M.A. and Wright, J.L. (Eds.), Feathered Dragons: Studies on the Transition from Dinosaurs to Birds, Indiana University Press, Indianapolis, IN, pp. 67–111, 2004. Return to text.
- Nesbitt, S.J., Turner, A.H., Spaulding, M., Conrad, J.L. and Norell, M.A., The theropod furcula, Journal of Morphology 270:856–879, 2009. Return to text.
- Norell, M.A., Makovicky, P. and Clark, J.M., A Velociraptor wishbone, Nature 389:447, 1997. Return to text.
- Tykoski, R., Forster, C.A., Rowe, T., Sampson, S.D. and Munyikwa, D., A furcula in the coelophysid theropod Syntarsus, Journal of Vertebrate Paleontology 22(3):728–733, 2002. Return to text.
- Yates, A.M. and Vasconcelos, C.C., Furcula-like clavicles in the prosauropod dinosaur Massospondylus, Journal of Vertebrate Paleontology 25(2):466–468, 2005. Return to text.
- Chiappe, ref. 1, p. 34. Return to text.
- Barrett, ref. 14, pp. 64, 117. Return to text.
- Chiappe, ref. 1, pp. 1–268. Return to text.
- Currie, P.J. and Chen, P.-J., Anatomy of Sinosauropteryx prima from Liaoning, northeast China, Canadian Journal of Earth Science 38:1705–1727, 2001. Return to text.
- Norell, M.A. and Xu, X., Feathered dinosaurs, Annual Review of Earth and Planetary Science 33:277–299, 2005. Return to text.
- Zhou, Z., Barrett, P.M. and Hilton, J., An exceptionally preserved lower Cretaceous ecosystem, Nature 421:807–814, 2003. Return to text.
- Xu, X., Wang, X.-L. and Wu, X.-C., A dromaeosaurid dinosaur with a filamentous integument from the Yixian Formation of China, Nature 401:262–266, 1999. Return to text.
- Stone, R., Bird-feather link firmed up, and in brilliant Technicolor, Science 327:508, 2010. Return to text.
- Zhang, F., Kearns, S.L., Orr, P.J., Benton, M.J., Zhou, Z., Johnson, D., Xu, X. and Wang, X., Fossilized melanosomes and the colour of Cretaceous dinosaurs and birds, Nature 463:1075–1078, 2010. Return to text.
- Xu, X., Zhou, Z., Wang, X., Kuang, X., Zhang, F. and Du, X., Four-winged dinosaurs from China, Nature 421:335–340, 2003. Return to text.
- Sereno, P.C., The origin and evolution of dinosaurs, Annual Review of Earth and Planetary Science 25:459, 1997. Return to text.
- Currie, P.J., Theropods, including birds; in: Currie, P.J. and Koppelhus E.B. (Eds.), Dinosaur Provincial Park: A Spectacular Ancient Ecosystem Revealed, Indiana University Press, Indianapolis, IN, p. 367, 2005. Return to text.
- Currie, ref. 31, pp. 367–397. Return to text.
- Prum, R.O., Are current critiques of the theropod origin of birds science? Rebuttal to Feduccia (2002), The Auk 120(2):550–561, 2003. Return to text.
- Prum, R.O., Who’s your daddy? Science 322:1799, 2008. Return to text.
- Varricchio, D.J., Moore, J.R., Erickson, G.M., Norell, M.A., Jackson, F.D. and Borkowski, J.J., Avian paternal care had dinosaur origin, Science 322:1826–1828, 2008. Return to text.
- Feduccia, A., Martin, L.D. and Simmons, J.E., Nesting dinosaur, Science 272:1571, 1996 Return to text.
- Varricchio, D.J. and Jackson, F.D., Two eggs sunny-side up: reproductive physiology in the dinosaur Troodon formosus; in: Currie, P.J., Koppelhus, E.B., Shugar, M.A. and Wright J.L. (Eds.), Feathered Dragons: Studies on the Transition from Dinosaurs to Birds, Indiana University Press, Indianapolis, IN, pp. 215–233, 2004. Return to text.
- Feduccia, ref. 2, p. viii. Return to text.
- Stokstad, E., Tiny, feathered dino is most birdlike yet, Science 290:1871–1872, 2000. Return to text.
- Graham-Kennedy, E., Dinosaurs: Where Did They Come from … and Where Did They Go? Pacific Press Publishing Association, Nampa, ID, 2006. Return to text.
- Barrett, ref. 14, p. 61. Return to text.
- DeCourten, F., Dinosaurs of Utah, The University of Utah Press, Salt Lake City, UT, p. 29, 1998. Return to text.
- Feduccia, A., Lingham-Soliar, T. and Hinchliffe, J.R., Do feathered dinosaurs exist? Testing the hypothesis on neontological and paleontological evidence, Journal of Morphology 266:125–166, 2005. Return to text.
- Gibbons, A., Plucking the feathered dinosaur, Science 278:1229, 1997. Return to text.
- Mayr, G., Peters, D.S., Plodowski, G. and Vogel, O., Bristle-like integumentary structures at the tail of the horned dinosaur Psittacosaurus, Naturwissenschaften 89:361–365, 2002. Return to text.
- Lingham-Soliar, T., Feduccia, A. and Wang, X., A new Chinese specimen indicates that ‘protofeathers’ in the Early Cretaceous theropod dinosaur Sinosauropteryx are degraded collagen fibres, Proceedings of the Royal Society B 274:1823–1829, 2007. Return to text.
- Lingham-Soliar, T., A unique cross section through the skin of the dinosaur Psittacosaurus from China showing a complex fibre architecture, Proceedings of the Royal Society B 275:775–780, 2008. Return to text.
- Lingham-Soliar, T. and Plodowski, G., The integument of Psittacosaurus from Liaoning Province, China: taphonomy, epidermal patterns and color of a ceratopsian dinosaur, Naturwissenschaften 97:479–486, 2010. Return to text.
- Witmer, L.M., Fuzzy origins for feathers, Nature 458:293–295, 2009. Return to text.
- Zheng, X.-T., You, H.-L., Xu, X. and Dong, Z.-M., An early Cretaceous heterodontosaurid dinosaur with filamentous integumentary structures, Nature 458:333–336, 2009. Return to text.
- Feduccia, A., Birds and dinosaurs: simple answer to a complex problem, Auk 119(4):1187–1201, 2002. Return to text.
- Lingham-Soliar, T., The dinosaurian origin of feathers: perspectives from dolphin (Cetacea) collagen fibers, Naturwissenschaften 90:563–567, 2003. Return to text.
- Witmer, ref. 49, p. 295. Return to text.
- Lingham-Soliar, T., The evolution of the feather: Sinosuaropteryx, a colourful tail, Journal of Ornithology, published online 3 December 2010. Return to text.
- Lingham-Soliar, T., Dinosaur protofeathers: pushing back the origin of feathers into the Middle Triassic? Journal of Ornithology 151:199, 2010. Return to text.
- Feduccia, A., Martin, L.D. and Tarsitano, S., Perspectives in ornithology and Archaeopteryx 2007: Quo Vadis? Auk 124(2):273–280, 2007. Return to text.
- Brochu, C.A. and Norell, M.A., Temporal congruence and the origin of birds, Journal of Vertebrate Paleontology 20(1):197–200, 2000. Return to text.
- Hu, D., Hou, L., Zhang, L. and Xu, X., A pre-Archaeopteryx troodontid theropod from China with long feathers on the metatarsus, Nature 461:640–643, 2009. Return to text.
- Witmer, L.M., Feathered dinosaurs in a tangle, Nature 461:601–602, 2009. Return to text.
- Xu, S. and Zhang, F., A new maniraptoran dinosaur from China with long feathers on the metatarsus, Naturwissenschaften 92:173–177, 2005. Return to text.
- Choiniere, J.N., Xu, X., Clark, J.M., Forster, C.A., Guo, Y. and Han, F., A basal Alvarezsauroid theropod from the early late Jurassic of Xinjiang, China, Science 327:571–574, 2010. Return to text.
- Hu et al., ref. 58, p. 642. Return to text.
- Feduccia, ref. 51, p. 1197. Return to text.
- Göhlich, U.B. and Chiappe, L.M., A new carnivorous dinosaur from the Late Jurassic Solnhofen archipelago, Nature 440:329–332, 2006. Return to text.
- Xu, X., Scales, feathers and dinosaurs, Nature 440:287, 2006. Return to text.
- Doyle, S., Plucking the dinobird, creation.com/plucking-the-dinobird, 28 September 2007. Return to text.
- Doyle, S., ‘Jurassic Park’ feathers? creation.com/jurassic-park-feathers, 2 October 2007. Return to text.
- O’connor, P.M. and Claessens, L.P.A.M., Basic avian pulmonary design and flow-through ventilation in non-avian theropod dinosaurs, Nature 436:253–256, 2005. Return to text.
- Sereno, P.C., Martinez, R.N., Wilson, J.A., Varricchio, D.J., Alcober, O.A. and Larsson, H.C.E., Evidence for avian intrathoracic air sacs in a new predaroty dinosaur from Argentina, PLoS ONE 3(9):1–20, 2008. Return to text.
- Wieland, C., Dinos breathed like birds? Journal of Creation 19(3):11–12, 2005. Return to text.
- Ruben, J.A., Jones, T.D., Geist, N.R. and Hillenius, W.J., Lung structure and ventilation in theropod dinosaurs and early birds, Science 278:1267–1270, 1997. Return to text.
- Ruben, J.A., Sasso, C.D., Geist, N.R., Hillenius, W.J., Jones, T.D. and Signore, M., Pulmonary function and metabolic physiology of theropod dinosaurs, Science 283:514–516, 1999. Return to text.
- Gibbons, A., Lung fossils suggest dinos breathed in cold blood, Science 278:1230, 1997. Return to text.
- Quick, D.E. and Ruben, J.A., Cardio–pulmonary anatomy in theropod dinosaurs: implications from extant archosaurs, Journal of Morphology 270:1244, 2009. Return to text.
- Chiappe, ref. 1, pp. 52–54. Return to text.
- Ruben, J.A., Hillenius, W.J., Geist, N.R., Leitch, A., Jones, T.D., Currie, P.J., Horner, J.R. and Espe III, G., The metabolic status of some late Cretaceous dinosaurs, Science 273:1204–1207, 1996. Return to text.
- Burke, A.C. and Feduccia, A., Developmental patterns and the identification of homologies in the avian hand, Science 278:666–668, 1997. Return to text.
- Hinchliffe, R., The forward march of the bird-dinosaurs halted? Science 278:596–597, 1997. Return to text.
- Oard, M.J., Bird-dinosaur link challenged, Journal of Creation 12(1):5–7, 1998. Return to text.
- Feduccia, A. and Nowicki, J., The hand of birds revealed by early ostrich embryos, Naturwissenschaften 89:391–393, 2002. Return to text.
- Galis, F., Kundrát, M. and Sinervo, B., An old controversy solved: bird embryos have five fingers, Trends in Ecology and Evolution 18(1):7–9, 2003. Return to text.
- Wagner, G.P. and Gauthier, J.A., 1,2,3 = 2,3,4: a solution to the problem of the homology of the digits in the avian hand, Proceedings of the National Academy of Science 96:5111–5116, 1999. Return to text.
- Wagner and Gauthier, ref. 82, p. 5113. Return to text.
- Xu et al., A Jurassic ceratosaur from China helps clarify avian digital homologies, Nature 459:940–944, 2009. Return to text.
- James, F.C. and Pourtless IV, J.A., Cladistics and the origin of birds: a review and two new analyses, Ornithological Monographs 66:28, 2009. Return to text.
- Chiappe, ref. 1, pp. 54–58. Return to text.
- Feduccia, ref. 2, pp. 1–466. Return to text.
- Feduccia et al., ref. 56, p. 378. Return to text.
- Jones, T.D., Farlow, J.O., Ruben, J.A., Henderson, D.M. and Hillenius, W.J., Cursoriality in bipedal archosaurs, Nature 406:716–718, 2000. Return to text.
- Chiappe, ref. 1, p. 63. Return to text.
- Wellnhofer, P., The plumage of Archaeopteryx: feathers of a dinosaur? in: Currie, P.J., Koppelhus, E.B., Shugar, M.A. and Wright J.L. (Eds.), Feathered Dragons: Studies on the Transition from Dinosaurs to Birds, Indiana University Press, Indianapolis, IN, p. 283, 2004. Return to text.
- James and Pourtless, ref. 85, pp. 1–78. Return to text.
- Feduccia et al., ref. 43, p. 137. Return to text.
- Chen, P.-J., Dong, Z.-M.and Zhen, S.-N., An exceptionally well-preserved theropod dinosaur from the Yixian Formation of China, Nature 391:147–152, 1998. Return to text.
- Feduccia et al., ref. 56, p. 377. Return to text.
- Xu, X., Zhao, Q., Norell, M., Sullivan, C., Hone, D., Erickson, G., Wang, X., Han, F. and Guo, Y., A new feathered maniraptoran dinosaur fossil that fills a morphological gap in avian origin, Chinese Science Bulletin 54(3):430–435, 2009. Return to text.
- Feduccia, ref. 2, pp. 18–35. Return to text.
- Feduccia, ref. 2, p. 1. Return to text.
- Wellnhofer, ref. 91, p. 286. Return to text.
- Mayr, G., Pohl, B. and Peters, D.S., A well-preserved Archaeopteryx specimen with theropod features, Science 310:1483–1486, 2005. Return to text.
- Mayr, G., Pohl, B., Hartman, S. and Peters, D.S., The tenth skeletal specimen of Archaeopteryx, Zoological Journal of the Linnean Society 149:97–116, 2007. Return to text.
- Gish, D.T., Evolution: The Fossils Still Say No! Institute for Creation Research, Dallas, TX, pp. 129–145, 1995. Return to text.
- Barrett, ref. 14, pp. 158, 166. Return to text.
- Currie, ref. 31, p. 282. Return to text.
- Feduccia, ref. 2, p. 101. Return to text.
- Gish, ref. 102, p. 138. Return to text.
- Feduccia, A., Evidence from claw geometry indicating arboreal habits of Archaeopteryx, Science 259:790–793, 1993. Return to text.
- Morell, V., Archaeopteryx: early bird catches a can of worms, Science 259:764–765, 1993. Return to text.
- Oard, M.J., Archaeopteryx was a bird, Creation Research Society Quarterly 30:16, 1993. Return to text.
- Alonso, P.D., Milner, A.C., Ketcham, R.A., Cookson, M.J. and Rowe, T.B., The avian nature of the brian and inner ear of Archaeopteryx, Nature 430:666–669, 2004. Return to text.
- Speakman, J.R. and Thomson, S.C., Flight capabilities of Archaeopteryx, Nature 370:514, 1994. Return to text.
- Speakman J.R. and Thomson, S.C., Feather asymmetry in Archaeopteryx, Nature 374:221–222, 1995. Return to text.
- Alonso et al., ref. 110, p. 666. Return to text.
- Longrich, N., Structure and function of hindlimb feathers in Archaeopteryx lithographica, Paleobiology 32(3):417–431, 2006. Return to text.
- Norberg, R.A., Feather asymmetry in Archaeopteryx, Nature 374:221, 1995. Return to text.
- Chatterjee, S. and Templin, R.J., The flight of Archaeopteryx, Naturwissenschaften 90:29, 2003. Return to text.
- Wellnhofer, ref. 91, pp. 282–300. Return to text.
- Feduccia, ref. 107, p. 792. Return to text.
- Feduccia, ref. 2, p. 113. Return to text.
- Chiappe, ref. 1, p. 59. Return to text.
- Burke and Feduccia, ref. 77, p. 666. Return to text.
- Witmer, ref. 59, p. 601. Return to text.
- James and Pourtless, ref. 85, p. 1. Return to text.
- Oard, ref. 79, p. 7. Return to text.
- Jenner, R.A., The scientific status of metazoan cladistics: why current research practice must change, Zoologica Scripta 33(4):293–310, 2004. Return to text.
- Jenner, ref. 125, p. 293. Return to text.
- Burke and Feduccia, ref. 77, p. 668. Return to text.
- Fedducia, ref. 51, p. 1192. Return to text.
- Stokstad, E., Feathers, or flight of fancy? Science 288:2125, 2000. Return to text.
- Feduccia et al., ref. 43, p. 158. Return to text.
- Feduccia, ref. 2, p. 56 Return to text.





Readers’ comments
Comments are automatically closed 14 days after publication.