The collapse of ‘geologic time’

Tiny halos in coalified wood tell a story that demolishes ‘long age’.
The age of things is crucial in the debate over the authority of the Bible.
Most methods that could be used for calculating the Earth’s age, even though still based on unprovable uniformitarian1 assumptions, give upper limits much less than the billions of years required for evolution.2 Evolutionists widely use radio–isotope (or radiometric) dating of rocks to support the ‘geologic time’ figure of 4.6 billion years. Notwithstanding the inherent unreliability and demonstrated inaccuracy of the radiometric dating techniques (see ‘Radiometric dating’ box below), ages of rock formations in the millions (and billions) of years are presented as fact in schools, universities and the media.
However, there is spectacular, but little-known, evidence that is completely inconsistent with the evolutionary timescale, but entirely consistent with the Biblical record of a young Earth and a catastrophic global Flood.
The evidence is provided by radio–halos in coalified wood. This work has been published in some of the best peer-reviewed scientific journals, and its strong case against evolution’s millions of years is so far unanswered by the evolutionary community.
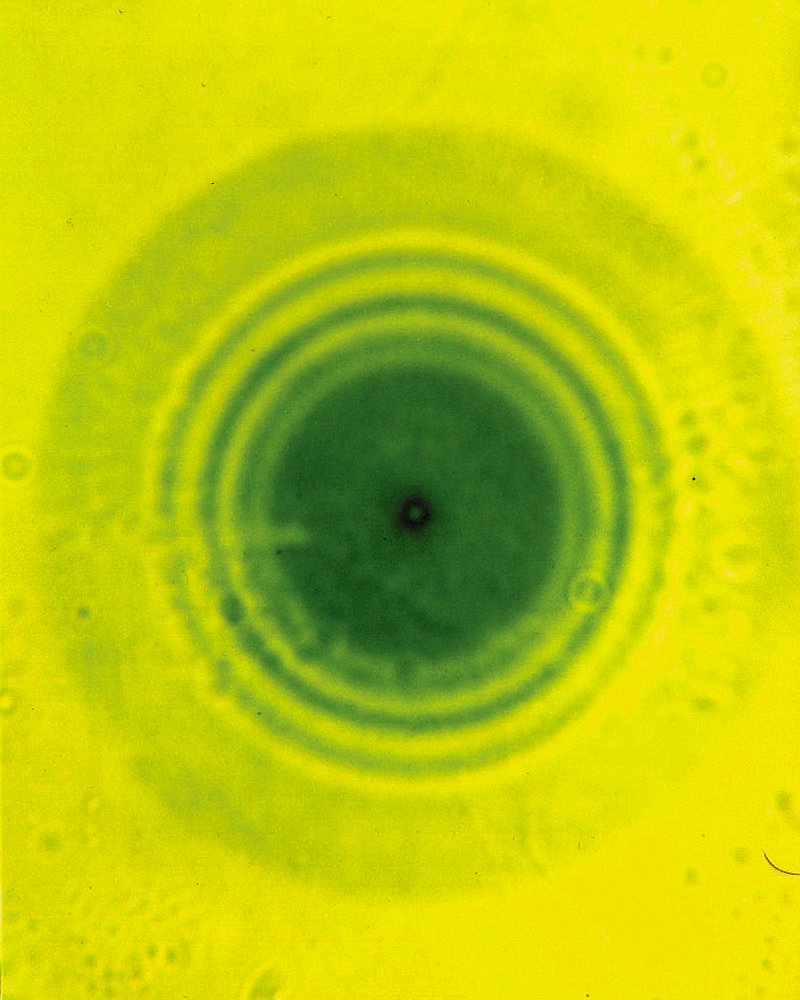


What are radiohalos?
Radiohalos are spherical, microscopic-sized discolourations in crystals. They are found abundantly in certain minerals in Earth’s rocks, especially micas from granites. In cross-section on a microscope slide, they appear as a series of tiny concentric rings, usually surrounding a central core (Figure 1).3
This central core is (at least initially) radioactive. High energy alpha particles, emitted from the core during radioactive decay, damage the mineral and discolour it, with most of the damage occurring where the particle stops. How far this particle travels depends on its energy. Since all the alpha particles from a particular type of decay reaction have the same energy, and the particles are fired in all directions, a spherical shell of discolouration will form, appearing circular in cross-section.
Imagine shooting a bullet into a huge lump of cork. Eventually, the bullet will stop, leaving behind a ‘trail’ of damage, the length of which depends on the speed of the bullet. Different radioactive substances shoot alpha particles (‘bullets’) at different (though specific) speeds, so we can identify the substance from the diameter of the ‘sphere of damage’.4 The higher the energy of decay, the faster the speed of the ‘bullet’.
Uranium radiohalos
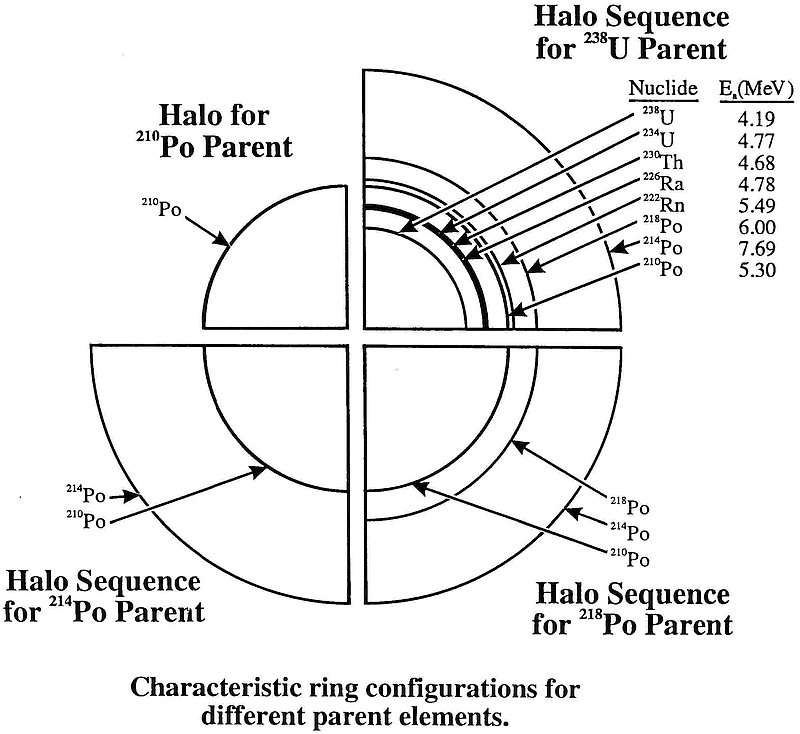
Radioactive uranium generates a beautiful, multi-ringed halo (Figure 1) because it decays in a number of steps. Of the 15 isotopes (or varieties of elements) in this ‘decay chain’ (see ‘Radioactive decay series’ box below), eight emit alpha particles when they decay, forming eight rings.5 It is a bit like a sequence of guns, each of different power, firing an eight-gun salute. When this salute or decay chain is fired millions of times in every direction, the bullets from the different guns make eight concentric rings.
If, instead of radioactive uranium, the core was composed of an isotope along the chain, there would be fewer rings. Omitting the first few isotopes in the decay series would be like removing the first few guns in our ‘salute’. Thus it’s quite simple to work out which isotope was originally in the core by counting the rings (see box below). Polonium-218 forms three rings, polonium-214 forms two, and polonium-210 forms only one.
Radiohalos in coalified wood
Radiohalos have also been found in logs recovered from uranium mines on the Colorado Plateau of Western USA. The logs, partially turned to coal, were found in uranium-rich sedimentary rocks from three different geological formations.
Some of these formations had previously been assigned radiometric ‘dates’ ranging from 55 to 80 million years.6 Scientists Jedwab7 and Breger8 described these halos, and Dr Robert Gentry, a world authority on radiohalos, revisited their work. Following extensive investigation, Gentry published his results in the prestigious journal Science,9 in a book10 and in a video.11
Most of the halos found in the wood had only one ring, indicating that the radioactive cores once contained polonium-210—the last radioactive isotope in the uranium-238 decay chain (see Radioactive decay series). Clearly, the wood had been saturated in uranium-rich solutions, and certain spots attracted polonium atoms (also present in these solutions), allowing small cores of polonium-210 to form. As they decayed, these cores left the characteristic polonium-210 halo.
Radioactive decay series
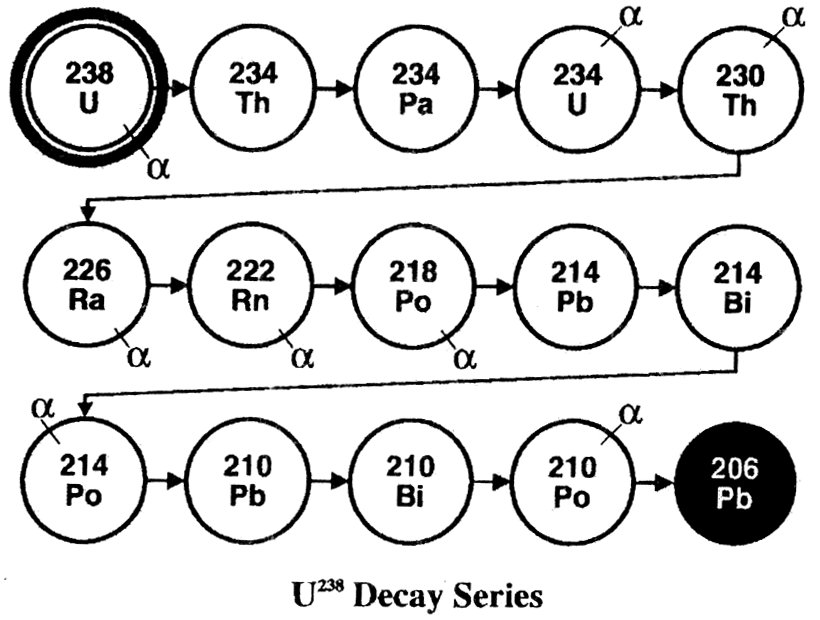
Radioactive isotopes have an intrinsically unstable atomic structure which makes them disintegrate so that particles fly out. One way that a parent radio-active atom can decay into a daughter atom is by ejecting an alpha particle from its nucleus. Sometimes the daughter element is also unstable and subsequently decays into another unstable isotope, and so on in a series of steps—a ‘decay chain’.
The isotope uranium-238 starts a decay chain that disintegrates step-by-step into a stable form of lead. It involves fifteen isotopes and fourteen steps (diagram right). Different isotopes of the same element (e.g. uranium-238 and uranium-235) have a different mass but nearly identical chemical behaviour. An alpha particle is a helium nucleus with a mass of 4 atomic mass units. Thus, radioactive decay by emission of an alpha particle (e.g. uranium-238) produces a daughter isotope (thorium-234) which is 4 atomic mass units lighter.
The half-life of a radioactive isotope is the time required for half its atoms to decay. Different isotopes have different half lives (e.g. the half-life of uranium-238 is 4.5 billion years and of polonium-218 is 3 minutes).
But the solutions must have penetrated the logs relatively quickly, certainly within a year or so. How do we know that? Because the half-life of polonium-210 is only 138 days. That is, within 138 days, half the polonium-210 present would have decayed into the next ‘daughter’ isotope in the chain. In other words, the solution had saturated the wood within two or three half-lives, about a year. It could not have taken very long, because in 10 half-lives (less than four years) virtually all of the polonium-210 would have gone.
Only one of the three radioactive isotopes of polonium was deposited in the tiny radioactive specks in the logs. We know because only one ring formed. The other isotopes from the decay chain (polonium-214 and polonium-218) were missing. Why? Because they had already decayed away. Their half-lives are very short (164 millionths of a second and three minutes respectively). So all polonium-214 would have disappeared within a thousandth of a second, and all polonium-218 would have gone in an hour—long before the uranium-rich solutions could saturate the logs.
Significantly, the halos were mainly elliptical, not circular (Figure 2). Obviously, after the halos formed, the wooden logs were compressed, squashing the originally-circular halos into ellipses.
Sometimes a circular halo could be seen together with an elliptical halo (Figure 3). This indicated that radioactive polonium-210 continued to decay from the same core after the wood was compressed. Thus, because of the 138-day half-life of polonium-210 as discussed above, there was less than four years between when the solution first infiltrated the wood and when it was compressed. (The presence of the second halo at the same spot shows that much less than four years had passed before the compression event, as there was still time to produce another halo afterwards.)12
An amazing event
The wood in which these tiny elliptical halos were found speaks of a devastating flood that uprooted and smashed huge trees, depositing the debris with an enormous volume of sediment over a large area. The halos themselves tell the story of an unusual geologic event. They speak of uranium-rich solutions saturating the logs in less than a year or so, forming tiny specks of polonium, which decayed to produce circular radiohalos, which were, in much less than four years, compressed and deformed.
The story is one of exceptional geological conditions—a highly unusual sequence of events. For one thing, in the usual ‘slow and gradual’ scenarios, it would take much longer for sufficient sediment to accumulate on top to deform the wood in this way. What is really amazing and significant, however, is the fact that this elliptical halo situation has been found in three different geological formations in the same general region. Evolutionists say these formations represent three different geological periods ranging from 35 to 245 million years.13 To believe this millions-of-years timescale, we would need to believe that this amazing sequence of events (with all its precise timing) occurred three different times, separated by more than 200 million years. Clearly this is an incredible scenario. It makes more sense to believe that the sequence occurred once and that all the sedimentary formations were deposited in the same catastrophe, followed by the same earth movement causing deformation. These polonium halos collapse the ‘long ages’ of geology, and point to the unique, catastrophic Flood recorded in the Bible. Also, by the same reasoning, these halos leave little room for numerous layers of post-flood sedimentation as suggested by some authors.14
Counting the rings

More confirming evidence
Further confirmation of this spectacular collapse of geologic time is provided by careful analysis of the tiny cores of some uranium halos found in the same wood samples.15 This revealed a large amount of uranium-238 but almost no lead-206.16 If the halos were millions of years old, much more ‘daughter’ lead should have been present. The scarcity of the daughter element, using the same assumptions upon which radiometric dating is based, would indicate that the halos are only several thousand years old, not millions. Similar results were obtained for halos from all three geological formations, indicating that all are approximately the same age. Again, the supposed millions of years of geologic time collapse into only a few thousand.17

Dinosaur tracks
Fossilized dinosaur footprints have been found in these Colorado mines. In Cyprus Plateau Mine (Utah), a fossilized dinosaur footprint was found in the coal seam next to one of the many coalified logs of the plateau. In Kenilworth Mine, eight different types of dinosaur tracks were found.
The pattern of tracks suggests that the animals were fleeing from an imminent catastrophe. Nearby, a huge dinosaur graveyard has been found at Dinosaur National Monument (Vernal, Utah) in Jurassic sediments.
Obviously, the dinosaurs that made these tracks didn’t escape. The catastrophe got them. The collapse of geologic time and the young age for the rock formations confirm that these dinosaurs lived on Earth, at the same time as man, only a few thousand years ago.
Radiometric dating relies on assumptions
Radiometric dating relies on three unprovable assumptions about the past:
- The amount of ‘daughter’ isotope in the rock at the start is known.
- No loss of ‘parent’ or gain of ‘daughter’ since the rock formed (closed system conditions).
- Constant decay rate of ‘parent’ to ‘daughter’.
If these conditions could be guaranteed, the radiometric dating method would be correct. However, unless eye–witnesses observed the rock when it formed, and checked it constantly thereafter, it is impossible to guarantee that these assumptions are correct. Indeed, there are many cases in the scientific literature where assumptions one and two, though made in good faith, have been shown to be unreliable.
Constancy of decay rate (assumption three) implies that a parameter which scientists have been measuring for only a century has been constant for millions of alleged years of Earth’s history. This is of course not only unproven but also unprovable. Decay rates (which can vary greatly today under special conditions) may have been much faster in the past; evidence suggesting this is now being analyzed by a creationist consortium.1 A good summary of the documented inconsistencies and inaccuracies of radiometric dating is given by Woodmorappe.2
- Vardiman, L., Snelling, A.A. and Chaffin, E.F. (Eds.), Radioisotopes and the Age of the Earth: A Young-Earth Creationist Research Initiative, Institute for Creation Research, California, and Creation Research Society, St. Joseph, Mississippi, 2000.
- Woodmorappe, J., The Mythology of Modern Dating Methods, Institute for Creation Research, California, 1999.
Conclusion
This scientific evidence, presented in leading journals, is a major problem for the idea of ‘millions of years’. It is, however, consistent with the vast fossil-bearing, sedimentary rock deposits of the Colorado Plateau having been laid down rapidly by the catastrophic global Flood described in the Bible, some 4,300 years ago. The dinosaurs that left footprints on the plateau, and were then buried and fossilized in the nearby rocks, also lived then—at the same time as man.
References and notes
- Such as the assumption of constant rates of change. Return to text.
- E.g. the amount of helium in the atmosphere, the decay and rapid reversals of Earth’s magnetic field, the salinity of the oceans, lack of continental erosion, and population statistics. A good summary is given by Morris, J.D., The Young Earth, Revised & Expanded, Master Books, Arizona, 1994. Return to text.
- Primary polonium-218 radiohalos command attention because they provide a record of extinct radioactivity in minerals constituting some of Earth’s most ancient rocks. See Gentry, R.V., Creation’s Tiny Mystery (3rd ed.), Earth Science Associates, Tennessee, 1992. Return to text.
- Obviously not a perfect analogy, despite being useful—a bullet into cork leaves equivalent damage all along its path, unlike the alpha particles which do most damage at the end, as stated. Return to text.
- The others decay by beta decay (β), not alpha (α). Note that due to overlap, only five of the eight rings of a 238U halo are normally visible. Return to text.
- Steiff, L.R. et al., A preliminary determination of the age of some uranium ores of the Colorado Plateau by the lead-uranium method, US Geological Survey Circular 271, 1953. Return to text.
- Jedwab, J., in: Given, P. (Ed.), Coal Science, American Chemical Society, Washington D.C., 1966. Return to text.
- Breger, I., in: Formation of Uranium Ore Deposits, Proceedings of Symposium in Athens 6–10 May 1974, pp. 99–124, International Atomic Energy Authority, Vienna, 1974. Return to text.
- Gentry, R.V., et al., Radiohalos and coalified wood: new evidence relating to the time of uranium introduction and coalification, Science 194:315–318, 1976. Return to text.
- Ref. 3, pp. 51–62. Return to text.
- Gentry, R.V., The Young Age of the Earth, Earth Science Associates LLC, Alpha Productions, 1996. Return to text.
- Gentry points out that the second halo could have formed from the decay of an isotope two steps back along the ‘chain’. Since the intermediary isotope undergoes beta, not alpha decay, the two possibilities cannot be distinguished from the halo. But this would stretch things from a maximum of around four years to some 22 years; a trivial matter. Return to text.
- The Eocene (supposedly 35–55 million years ago), the Jurassic (140–205 million years ago) and the Triassic (205–245 million years ago). See ref. 3, p. 56. Return to text.
- See refs. in McIntosh, A.C., Edmondson, T. and Taylor, S., Genesis and catastrophe: the Flood as the major Biblical cataclysm, Journal of Creation 14(1):101–109, 2000. Return to text.
- Using X-ray fluorescence (EXMRF) and the more sensitive ion microprobe mass analysis (IMMA). Return to text.
- The ratios of uranium to lead were up to 64,000, indicating the halos are only thousands of years old. Halos millions of years old would have a far lower uranium-to-lead ratio. For details see ref. 3, pp. 61–62; and ref. 9. Return to text.
- It is chemically implausible to believe that lead could be leached out, leaving uranium—the reverse is far more likely. Return to text.


Readers’ comments
Comments are automatically closed 14 days after publication.